Posted on 07/16/2021 9:18:29 AM PDT by Red Badger

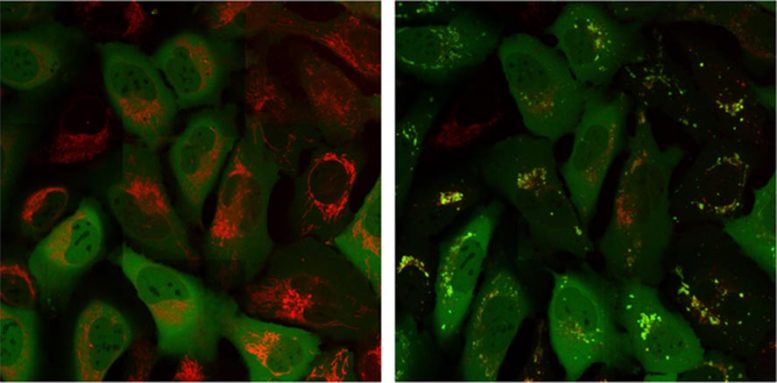
Parkin protein (green signal) is in a different part of the cell than the mitochondria (red signal) at time 0 (left image) but then co-localizes with the mitochondria after 60 minutes (right image). Credit: Salk Institute
==================================================================================
Enzyme with central role in cancer and type 2 diabetes also activates “clean-up” protein in Parkinson’s.
When cells are stressed, chemical alarms go off, setting in motion a flurry of activity that protects the cell’s most important players. During the rush, a protein called Parkin hurries to protect the mitochondria, the power stations that generate energy for the cell. Now Salk researchers have discovered a direct link between a master sensor of cell stress and Parkin itself. The same pathway is also tied to type 2 diabetes and cancer, which could open a new avenue for treating all three diseases.
“Our findings represent the earliest step in Parkin’s alarm response that anyone’s ever found by a long shot. All the other known biochemical events happen at one hour; we’ve now found something that happens within five minutes,” says Professor Reuben Shaw, director of the NCI-designated Salk Cancer Center and senior author of the new work, detailed in Science Advances on April 7, 2021. “Decoding this major step in the way cells dispose of defective mitochondria has implications for a number of diseases.”
Parkin’s job is to clear away mitochondria that have been damaged by cellular stress so that new ones can take their place, a process called mitophagy. However, Parkin is mutated in familial Parkinson’s disease, making the protein unable to clear away damaged mitochondria. While scientists have known for some time that Parkin somehow senses mitochondrial stress and initiates the process of mitophagy, no one understood exactly how Parkin was first sensing problems with the mitochondria—Parkin somehow knew to migrate to the mitochondria after mitochondrial damage, but there was no known signal to Parkin until after it arrived there.
Shaw’s lab, which is well known for their work in the fields of metabolism and cancer, spent years intensely researching how the cell regulates a more general process of cellular cleaning and recycling called autophagy. About ten years ago, they discovered that an enzyme called AMPK, which is highly sensitive to cellular stress of many kinds, including mitochondrial damage, controls autophagy by activating an enzyme called ULK1.
Following that discovery, Shaw and graduate student Portia Lombardo began searching for autophagy-related proteins directly activated by ULK1. They screened about 50 different proteins, expecting about 10 percent to fit. They were shocked when Parkin topped the list. Biochemical pathways are usually very convoluted, involving up to 50 participants, each activating the next. Finding that a process as important as mitophagy is initiated by only three participants—first AMPK, then ULK1, then Parkin—was so surprising that Shaw could scarcely believe it.
To confirm the findings were correct, the team used mass spectrometry to reveal precisely where ULK1 was attaching a phosphate group to Parkin. They found that it landed in a new region other researchers had recently found to be critical for Parkin activation but hadn’t known why. A postdoctoral fellow in Shaw’s lab, Chien-Min Hung, then did precise biochemical studies to prove each aspect of the timeline and delineated which proteins were doing what, and where. Shaw’s research now begins to explain this key first step in Parkin activation, which Shaw hypothesizes may serve as a “heads-up” signal from AMPK down the chain of command through ULK1 to Parkin to go check out the mitochondria after a first wave of incoming damage, and, if necessary, trigger destruction of those mitochondria that are too gravely damaged to regain function.
The findings have wide-ranging implications. AMPK, the central sensor of the cell’s metabolism, is itself activated by a tumor suppressor protein called LKB1 that is involved in a number of cancers, as established by Shaw in prior work, and it is activated by a type 2 diabetes drug called metformin. Meanwhile, numerous studies show that diabetes patients taking metformin exhibit lower risks of both cancer and aging comorbidities. Indeed, metformin is currently being pursued as one of the first ever “anti-aging” therapeutics in clinical trials.
“The big takeaway for me is that metabolism and changes in the health of your mitochondria are critical in cancer, they’re critical in diabetes, and they’re critical in neurodegenerative diseases,” says Shaw, who holds the William R. Brody Chair. “Our finding says that a diabetes drug that activates AMPK, which we previously showed can suppress cancer, may also help restore function in patients with neurodegenerative disease. That’s because the general mechanisms that underpin the health of the cells in our bodies are way more integrated than anyone could have ever imagined.”
Reference: “AMPK/ULK1-mediated phosphorylation of Parkin ACT domain mediates an early step in mitophagy” by Chien-Min Hung, Portia S. Lombardo, Nazma Malik, Sonja N. Brun, Kristina Hellberg, Jeanine L. Van Nostrand, Daniel Garcia, Joshua Baumgart, Ken Diffenderfer, John M. Asara and Reuben J. Shaw, 7 April 2021, Science Advances.
DOI: 10.1126/sciadv.abg4544
How about someone mansplain this?
At some time in the future, you may you forever because you will never get cancer, diabetes or Parkinson’s disease.
"Biochemical pathways are usually very convoluted, involving up to 50 participants, each activating the next. Finding that a process as important as mitophagy is initiated by only three participants—first AMPK, then ULK1, then Parkin—was so surprising that Shaw could scarcely believe it.Not only does the messaging instruct proteins to go check out mitochondrial damage, but the proteins can detect the extent of damage and decide which cells are too far gone and must be destroyed. All at the intracellular and intra-organelle level. That is so astonishing and mind-boggling! How humans can figure this out in that vanishingly small world with instrumentation is just incredible....Shaw’s research now begins to explain this key first step in Parkin activation, which Shaw hypothesizes may serve as a “heads-up” signal from AMPK down the chain of command through ULK1 to Parkin to go check out the mitochondria after a first wave of incoming damage, and, if necessary, trigger destruction of those mitochondria that are too gravely damaged to regain function.
Mitochondria are membrane-bound cell organelles that generate most of the chemical energy needed to power the cell's biochemical reactions. Chemical energy produced by the mitochondria is stored in a small molecule called adenosine triphosphate (ATP). Mitochondria contain their own small chromosomes. Generally, mitochondria, and therefore mitochondrial DNA, are inherited only from the mother.Mitochondria are membrane-bound with two different membranes which is quite unusual for an intercellular organelle. Those membranes function in the purpose of mitochondria, which is essentially to produce energy. That energy is produced by having chemicals within the cell go through pathways, in other words, be converted. And the process of that conversion produces energy in the form of ATP, because the phosphate is a high-energy bond and provides energy for other reactions within the cell. So the mitochondria's purpose is to produce that energy. Some different cells have different amounts of mitochondria because they need more energy. So for example, the muscle has a lot of mitochondria, the liver does too, the kidney as well, and to a certain extent, the brain, which lives off of the energy those mitochondria produce. So if you have a defect in the pathways that the mitochondria usually functions with, you're going to have symptoms in the muscle, in the brain, sometimes in the kidneys as well; many different types of symptoms. And we probably don't know all of the different diseases that mitochondrial dysfunction causes.
William Gahl, M.D., Ph.D. National Human Genome Research Institute
I think it is saying,,
The cells that intake energy get stressed, and a breakdown in parkins causes the broken down mitochondria to not be cleared away properly, which causes illness
Someone correct me if I’m wrong
bttt
The metabo,metabolism, system gets damaged, the body doesn’t get the required energy loads it needs to help fight off diseases as well as it did when the metabolic system was healthy
It’s kinda like a battery . At full charge, it can run powerful tools and such, but when the battery drains, you start to notice the tools aren’t as powerful, until finally they can’t do,the job they are capable of at full,charge
AGAIN, SOMEONE CORRECT. Me if I’m wrong, but thT is how I understand the breakdown to the mitochondria, which are the bodies energy powergouses
[[VParkin’s job is to clear away mitochondria that have been damaged by cellular stress so that new ones can take their place, a process called mitophagy. However, Parkin is mutated in familial Parkinson’s disease, making the protein unable to clear away damaged mitochondria. ]]
I’ll try again, the last reposnses I thjnk wer e nt the greatest
Think of the body running an a bunch of batteries that the boy constantly replenishes when they die or get weak. The Larkin somehow sense that the batteries have died, and come in and clear them so the body can replace them with new strong ones, enabling the body to keep on functioning at a high level to fight off diseases and such.
In Parkinson’s disease, the parkin can no longer clear put the old dead or dying batteries that are ‘stressed, so the body can’t put new strong healthy batteries in their place anmore, so the body gets weaker and weaker as more and more dead batteries accumulate. The body no longer has ‘full power to fight off diseases and whatnot
Something tips off the parkin that the batteries are dying. Researchers have zeroed in on what they think tips them off
Possibly metformin, a diabetes drug, may trigger something that initiates the parkin. To go in and clear, out the dead batteries so that new ones can be placed.
This ‘may mean’ longer life, less aging related diseases because the body has more energy because the mitochondria, the bodies powerhouse cells,, are healthy and more numerous now again, which enables them to energize the body so it can fight off diseases and such now.
There are a,ways problems iwth analogies, but I think this kinda sums up what is going on
Dang it. Autocorrect strikes Agatha ‘body’ constantly replaces,
And the ‘parkin’ somehow sense that the batteries have died, or rather that the mitochondria are damaged
Metformin seems promising, diabetics even live longer on it. Probably too cheap for big pharma to invest in clinical trials but it is already available OTC many places outside the US. I take NMN which also has an energizing effect on mitochondria.
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC7238909/
red light therapy ??
What is meant by “red light”?
Different wavelength or what?
Being a lowly mechanical engineer without a degree in organic chemistry, I could only understand things at the most superficial level.
Old people and men are at higher risk. They have more stored iron.
Diabetes can be caused by excess iron, maybe these conditions too.
People who drink coffee or take statins are at lower risk for the wuhan coronavirus. Drinking coffee or taking statins reduce iron absorption.
I know people on Metformin who now have Parkinson’s.
It may be helping beneath the surface where it isn’t apparent too sometimes health benefits aren’t noticed, but might help in the long run
Is it the coffee or the caffeine ?
People who drink coffee or take statins are at lower risk. -- That's me, too. Good news.
So I guess it's all a wash.
cellular damage activates
a tumor suppressor protein called LKB1,
which activates
an enzyne called AMPK, the central sensor of the cell’s metabolism,
which activates
an enzyme called ULK1,
which activates a protein called Parkin. by attaching a phosphate group to it.
Parkin then travels to damaged mitochondria and clears them away, so that new ones can take their place, a process called mitophagy.
Metformin can also activate LKB1, which explains metformin’s life extension and cancer prevention characteristics
As far as I know, in addition to metformin, low animal protein diet combined with exercise, calorie restriction/fasting, etc are the best activators of AMPK. Various other enzymes having similar synergistic effects, touching different parts of the elephant (just not as much in the dark anymore).
whats nmn?
Disclaimer: Opinions posted on Free Republic are those of the individual posters and do not necessarily represent the opinion of Free Republic or its management. All materials posted herein are protected by copyright law and the exemption for fair use of copyrighted works.