Prof. Raoult has done it. The French have a treatment. Raoult has eliminated the control group, and opened the trial to all.
Cuomo is going to run Raoult's protocols starting Tuesday in New York disguised as a 'massive trial' -- it's no trial, it's the actual treatment!
The FDA using Regeneron labs in New York, will provide Cuomo with 10,000 PQL+AZT units -- unironically nearly equal to the amount of cases in New York.
TEVA Pharms makes generics of both PQL and AZT and is donating 6 million doses to US starting now to the end of March. Azithromycin is not in short supply, and is not particularly hard to make (Israeli firm TEVA Pharms makes a generic of AZT too, as do about 10 other companies).
The French won the battle and maybe the war. Vive Didier Raoult! Vive La France!
Navigation: use the links below to view more comments.
first 1-20, 21-38 next last
To: StAnDeliver
Just WOW on PLQ + AZT. Incredible.
Can we lift the “shelter in place” order now?
To: StAnDeliver
Doctor Fauci and the bureaucratic CDC DO NOT APPROVE! ;-)
Props to these French doctors.
3 posted on
03/22/2020 10:28:52 AM PDT by
House Atreides
(It is not a HOAX it IS A PRETEXT!)
To: StAnDeliver
Great post. I’ve seen a lot of good articles lately, but yours is the best I’ve seen in a long time.
Thanks
5 posted on
03/22/2020 10:30:05 AM PDT by
be-baw
To: StAnDeliver
6 posted on
03/22/2020 10:30:47 AM PDT by
Yaelle
To: StAnDeliver
Trials in the field. They’ll get a huge amount of data and save lives.
8 posted on
03/22/2020 10:32:11 AM PDT by
headstamp 2
(There's a stairway to heaven, but there's also a highway to hell.)
To: StAnDeliver
“...TEVA Pharms makes generics of both PQL and AZT and is donating 6 million doses to US starting now to the end of March. Azithromycin is not in short supply, and is not particularly hard to make (Israeli firm TEVA Pharms makes a generic of AZT too, as do about 10 other companies)....”
***********************************
Mylan is reopening a shuttered West Virginia manufacturing facility and expects delivery of product beginning in early April. I’m pretty sure the facility is in Morgantown WV. They have necessary “raw materials” on hand for 50 million tabs. This is important because of the possibility of supply constraints on precursor stock.
http://newsroom.mylan.com/2020-03-19-Mylan-Ramps-Up-U-S-Manufacturing-of-Hydroxychloroquine-Sulfate-Tablets-to-Meet-Potential-COVID-19-Patient-Needs
I believe there are 7 to 8 CURRENT manufacturers of Hydroxychloroquine and more gearing up to manufacture.
10 posted on
03/22/2020 10:37:38 AM PDT by
House Atreides
(It is not a HOAX it IS A PRETEXT!)
To: StAnDeliver
Very good.Thanks for posting this.
11 posted on
03/22/2020 10:37:50 AM PDT by
Bigg Red
(WWG1WGA)
To: StAnDeliver
“TEVA Pharms makes generics of both PQL and AZT and is donating 6 million doses to US starting now to the end of March. Azithromycin is not in short supply, and is not particularly hard to make (Israeli firm TEVA Pharms makes a generic of AZT too, as do about 10 other companies).”
i appreciate you posting this information; i felt like a lone voice in the wilderness trying to cut through all of the noise of FR ignorance trying to post information like that ...
12 posted on
03/22/2020 10:42:22 AM PDT by
catnipman
(Cat Nipman: Vote Republican in 2012 and only be called racist one more time!)
To: StAnDeliver
I wish FR had a Like button. You’d get ten thousand of them, and that’s just from me!
13 posted on
03/22/2020 10:45:17 AM PDT by
Orosius
To: StAnDeliver
The Wusan triad is Fever, Cough and Shortness of Breath. It is now considered diagnostic of COVID-19 infection by the New York City Board of Health. If a patient has Fever and Cough, then the this therapeutic regimen should start before the third point of the triad ,Shortness of Breath, is reached. By then the lungs have already been compromised and the clinical outcome is in doubt. People are concerned that the quinolones and Zithromax prolong the QT interval on EKG and this may lead to serious or fatal arrhythmias. Many therapeutic agents prolong the QT interval. However if a person develops a pneumonia the right side pressures of the heart increase rapidly and while myocardial tissue itself is not a direct target of the virus, the increased strain on the heart due to hypoxemia and increased pressures can in itself lead to arrhythmias.
Quinolones and Zithromax may or may not work. However as of now its the best physicians can offer to patients ill with COVID-19. RNA viruses are bad actors and developing a vaccine is difficult. However until an effective and safe vaccine is developed, this virus will continue to devastate humans and their culture.
BTW believe data out of China at your own risk.
17 posted on
03/22/2020 10:47:29 AM PDT by
allendale
(.)
To: StAnDeliver
Merci, Professeur.
Professeur Didier Raoult, director of the Research Unit in Infectious and Tropical Emergent Diseases,
IHU Méditerranée Infection, Aix-Marseille University |Photo by Julien Poupart/ABACAPRESS.COM
21 posted on
03/22/2020 10:53:06 AM PDT by
StAnDeliver
(CNN's Dana B: "Show of hands: Coverage for undocumented immigrants?" ***all Democrat hands raised***)
To: StAnDeliver
PQL? PLQ? what do they mean?
22 posted on
03/22/2020 10:53:36 AM PDT by
ding_dong_daddy_from_dumas
(Mozart tells you what it's like to be human. Bach tells you what it's like to be the universe.)
To: StAnDeliver; All
The University of Minnesota is launching a clinical trial on a post-exposure treatment for coronavirus COVID-19 disease. They are recruiting test subjects.
COVID-19 Clinical Trial Launches at University of Minnesota
by Kelly Glynn, Media Relations Coordinator March 17, 2020
If you think you may be eligible to participate in the trial, please email covid19@umn.edu for further instructions.
The trial is being limited to only high-risk exposures, so that the trial may be completed as quickly as possible with the fewest number of volunteers. At this time, people who may have been exposed to coronavirus in the community are currently not eligible to participate.
To: StAnDeliver
The announcement does not make this an official protocol for all of France, just for this institute/hospital. Or am I missing something?
30 posted on
03/22/2020 11:03:09 AM PDT by
Chaguito
To: StAnDeliver
32 posted on
03/22/2020 11:24:50 AM PDT by
Hoosier-Daddy
("Washington, DC. You will never find a more wretched hive of scum and villainy. We must be cautious")
To: StAnDeliver
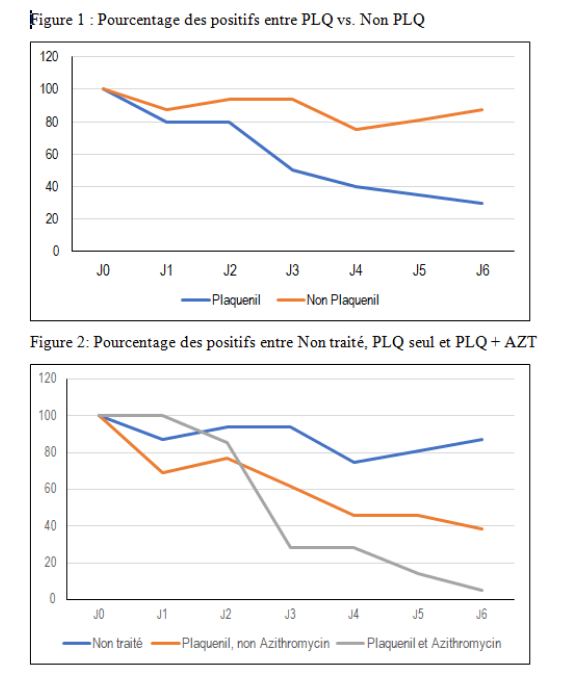
I don’t know how large their sample was, but it looks like the two drugs together have about a 95-97% cure rate.
To: Admin Moderator
How is not ExtendedNews, if not BreakingNews?
Was it reported as too difficult to understand? Perhaps too much French? Too many numbers? Multi-syllable words?
Cuomo is going to unleash this treatment on Tuesday ... would you rather wait for FoxNews to put out 5 grafs of short junk with the requisite grammatical errors?
41 posted on
03/22/2020 1:03:31 PM PDT by
StAnDeliver
(CNN's Dana B: "Show of hands: Coverage for undocumented immigrants?" ***all Democrat hands raised***)
To: StAnDeliver
To: StAnDeliver
WAIT A MINUTE....this has not been approved by Dr. Farci.
49 posted on
03/22/2020 4:24:33 PM PDT by
eartick
(Stupidity is expecting the government that broke itself to go out and fix itself. Texan for TEXIT!)
To: StAnDeliver
Glad Dr Fauxi was not in today’s briefing. He stands in the way of treatment protecting his turf and his Big Pharma buddies.
Navigation: use the links below to view more comments.
first 1-20, 21-38 next last
FreeRepublic.com is powered by software copyright 2000-2008 John Robinson

