Posted on 09/20/2020 9:49:37 PM PDT by BenLurkin
A research team from Caltech and the UCLA Samueli School of Engineering has demonstrated a promising way to efficiently convert carbon dioxide into ethylene — an important chemical used to produce plastics, solvents, cosmetics and other important products globally.
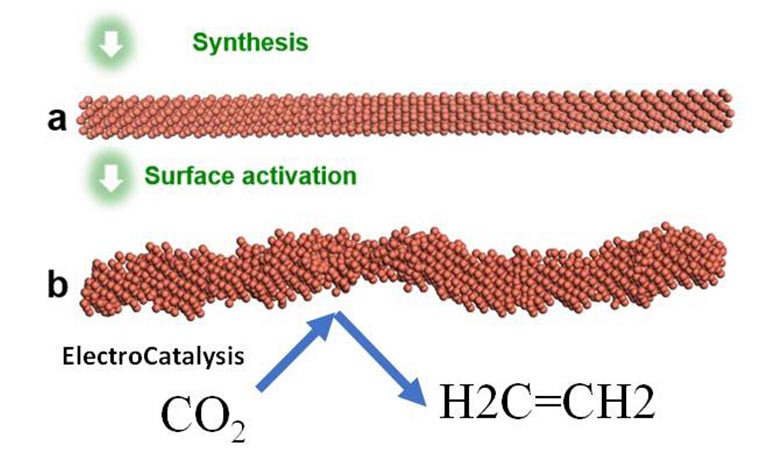
The scientists developed nanoscale copper wires with specially shaped surfaces to catalyze a chemical reaction that reduces greenhouse gas emissions while generating ethylene — a valuable chemical simultaneously. Computational studies of the reaction show the shaped catalyst favors the production of ethylene over hydrogen or methane. A study detailing the advance was published in Nature Catalysis.
Currently, ethylene has a global annual production of 158 million tons. Much of that is turned into polyethylene, which is used in plastic packaging. Ethylene is processed from hydrocarbons, such as natural gas.
Using copper to kick start the carbon dioxide (CO2) reduction into ethylene reaction (C2H4) has suffered two strikes against it. First, the initial chemical reaction also produced hydrogen and methane — both undesirable in industrial production. Second, previous attempts that resulted in ethylene production did not last long, with conversion efficiency tailing off as the system continued to run.
To overcome these two hurdles, the researchers focused on the design of the copper nanowires with highly active “steps” — similar to a set of stairs arranged at atomic scale. One intriguing finding of this collaborative study is that this step pattern across the nanowires’ surfaces remained stable under the reaction conditions, contrary to general belief that these high energy features would smooth out. This is the key to both the system’s durability and selectivity in producing ethylene, instead of other end products.

(Excerpt) Read more at scitechdaily.com ...
phew, just in time, the world was just about ready to burst into flames in 11 more years- /s
Whelp, here comes the ice age.
Regards,
Thank you!
Huh.
The anti-carbonists get their way with things and we can say bye bye to agriculture.
Crops need CO2 to flourish.
Ethylene production: Intellectually interesting, if it’s economical.
Should have gone for producing ethanol, liquor is quicker...
Using copper to kick start the carbon dioxide (CO2) reduction into ethylene reaction (C2H4) has suffered two strikes against it. First, the initial chemical reaction also produced hydrogen and methane — both undesirable in industrial production. Second, previous attempts that resulted in ethylene production did not last long, with conversion efficiency tailing off as the system continued to run.
So, I wonder, how much hydrogen, how much methane? Both can power fuel cells.
So we’re going to spend tons of money and use lots of energy to reduce a relatively infinitesimal amount of CO2 in the atmosphere, producing ethylene, “a valuable chemical” whose major use is the in the production of plastic. Sounds good to me.
That sure would be some expensive polyethylene made out of carbon dioxide.
Never mind the fact that they are way, way overstating the amount of carbon dioxide they claim is in the atmosphere.
i put the /s tag on- AOC says world gonna end in 10 years- due to CO2-
She’s nuts-
My thoughts exactly. The idea that we know enough to know what the appropriate level of CO2 is, to go monkeying with levels, seem extremely hubristic. We’ve had a quarter century of not funding anyone with contrary, heretical views on the subject.
“We are at the brink of fossil fuel exhaustion, coupled with global climate change challenges”
opening statements like that pretty much destroy any credibility for the rest of the article ...
Fake News. They lie by omission. They omitted the fact that this process requires energy, far more than was used when oxidizing the carbon. It takes more energy to create each ethylene molecule than was created by burning the Carbon that produced the CO2. Even with a 100 percent efficient catalyst, it would take just as much energy as was used in the burning. A catalyst can’t create energy.
Researchers Discover Nanoscale Catalyst to Efficiently Convert CO2 Into Ethylene
They’ve killed two birds with one stone: ridding the bounteous Earth of hated CO2 and ugly vegetation, while making fuel for cars to run on. Absolutely Brilliant!™
Crops need CO2 to flourish.
—
And we need the oxygen the crops produce to breathe
How about planting trees?
I got that. I didn’t mean to sound pedanticy.
It’s fun to get into leftists faces and scream at them about why they hate plants so much they want them all to suffocate.
Just because a chemical process takes a net energy input to create a desired end product doesn’t mean it’s not a valuable process. Depending on the source hydrocarbon a gallon of gasoline in its final RON 90 form takes anywhere from 1.25 to 1.5 gallons of gas equivalent in energy on a BTU in to BTU out basis. If the input energy is cheaper than the output products value it doesn’t matter what the energy balance is. Gasoline is hydrocracked using steam methane reformed hydrogen and process heat from much cheaper natural gas to make a much higher value liquid no one cares that cracking heavy visbreak or vaccum resid using anywhere from 25 to 75% of the final BTU value in the finished gallon in $1.50 or less per MMBTU in natural gas. All that matters is that the input energy costs are less than what the finished product will sell for. Propylene is a high value petrochemical if the cost of cheap natural gas or even nuclear energy is less per unit of CO2 cracked into finshed propylene then it doesn’t matter one hoot that the process consumes energy. Many MANY industrial processes consume massive amounts of energy to make a higher value end product. I’m a petroleum geologist and also a engineer by degrees we see every day at the refinery energy being consumed to make a end product.
Disclaimer: Opinions posted on Free Republic are those of the individual posters and do not necessarily represent the opinion of Free Republic or its management. All materials posted herein are protected by copyright law and the exemption for fair use of copyrighted works.