To: All
The language in the statute is as follows: " that there is no adequate, approved, and available alternative to the product for diagnosing, preventing, or treating such disease or condition" So the approved Comirnaty mRNA treatment must be both approved and available before the EUA must be revoked, and ti won't be available any time soon. You are missing a very important part of 360bbb–3. It's the piece of the puzzle that makes it all fit together. I know that you liked to play lawyer and pretend to be what you aren't so I'll give you a chance to find it.
As for the EUA, it is absolutely irrelevant. Pfizer/BioNTech has immunity whether the products are distributed pursuant to full approval or under a EUA. Your appeal to German law notwithstanding.
To: TexasGurl24
You're missing this.
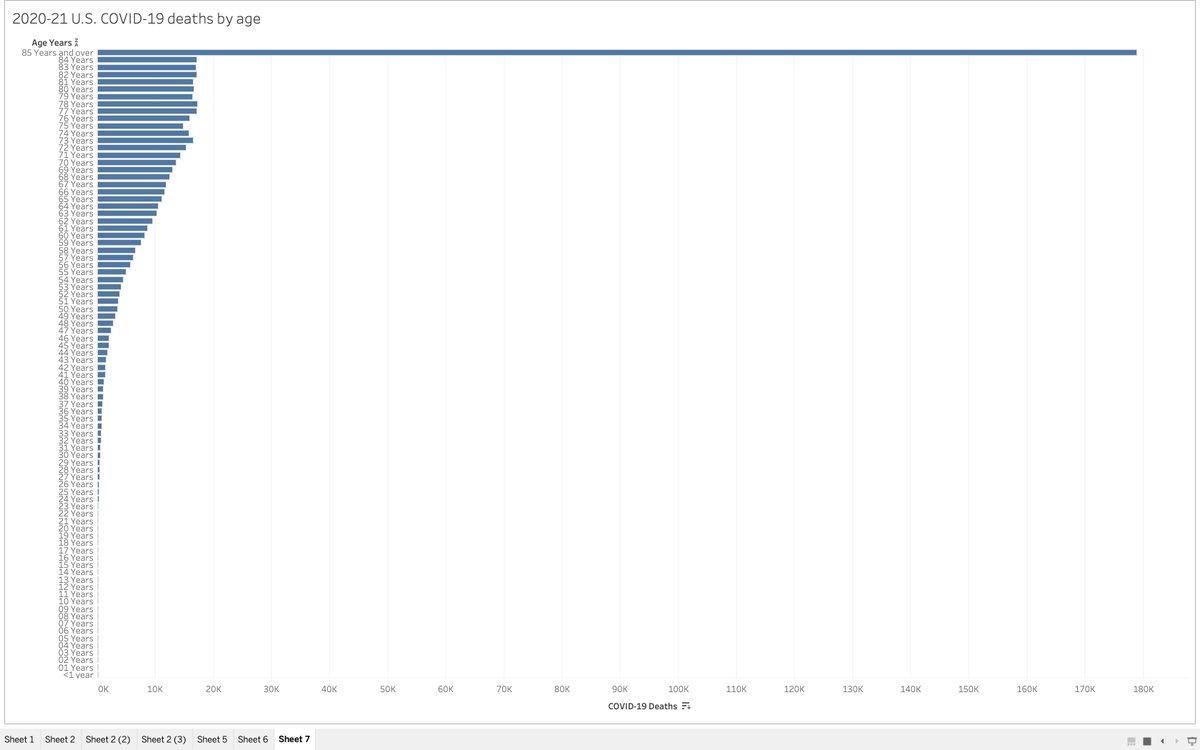
The whole thing is a sham.
23 posted on
08/26/2021 5:47:34 PM PDT by
grey_whiskers
(The opinions are solely those of the author and are subject to change with out notice.)
To: TexasGurl24
As for the EUA, it is absolutely irrelevant. Pfizer/BioNTech has immunity whether the products are distributed pursuant to full approval or under a EUA. Your appeal to German law notwithstanding.
The case law in Germany, from an American prosecutor and judge no less, is quite compelling:
https://www.fhi360.org/sites/all/libraries/webpages/fhi-retc2/Resources/nuremburg_code.pdf
46 posted on
08/26/2021 10:44:56 PM PDT by
Dr. Franklin
("A republic, if you can keep it." )
FreeRepublic.com is powered by software copyright 2000-2008 John Robinson

