To measure the date we use carbon dating. We also use many, many other methods, all based on wildly different methodologies, measurements, and premises. And yet, for the most part results of all the differently determined dating methods agree with each other. (And when they disagree, there are well-understood reasons why.) How do you explain this if you believe they are unreliable?
Furthermore, most items of evolutionary interest are *not* dated via "carbon dating", because Carbon-14 dating can only be used for items up to about 50,000 years old. Most items of evolutionary interest have ages measured in millions of years, and other methods are used. Carbon dating is primarily of use for items within the range of human history and early pre-history.
Yet can you answer with certainty the amount of carbon at any given time? As in, were the levels constant? The answer is no.
Very wrong. The answer is yes. There are many, many samples of known age (e.g. tree rings, arctic ice layers, lake bottom layers, etc.) which can be used to multiply and independently determine how much carbon-14 was in the atmosphere in any given year, and thus be used to calibrate Carbon-14 dating methods.
For a quick article on one such study, see http://more.abcnews.go.com/sections/science/dailynews/carbon0220.html
A much more technical treatment: Atmospheric Radiocarbon Calibration to 45,000 yr B.P.: Late Glacial Fluctuations and Cosmogenic Isotope Production
Such studies produce calibration results such as the following:
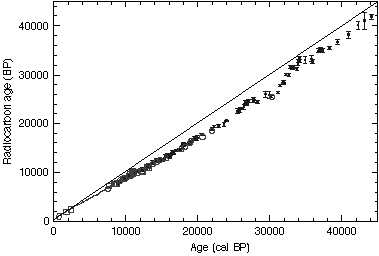
If the amount of Carbon-14 in the atmosphere had been exactly constant throughout time (and no one expects that it has been), then the results would fall on the straight diagonal line. Instead, the wiggly line indicates how much the actual amount of C-14 in the atmosphere deviated from the "base" amount, and from this we can know how much C-14 was actually present in any given year in the past 50,000 years.
Note that the above graph includes C-14 data from *two* completely independent sources (Lake Suigetsu varves, and ocean corals), and yet the results overlap beautifully, confirming each other. There is similar match from C-14 studies based on tree-ring data and other sources.
From this, we can build a Carbon-14 dating calibration or "correction" curve which can be used to confidently produce an accurate date from a given Carbon-14 measurement. These calibration curves look like this:

There are many databases available which are used to compile massive amounts of data to ensure the proper calibration of carbon-dating. For just one example, Marine Reservoir Correction Database.
Other methods are used to cross-check and calibrate other dating methods to ensure accuracy.
So we can get approximate dates, but relative to how close in terms of the universe?
Quite close.
If we can take a leaf from a tree and date it as being 10,000 years old, yet it was just removed and is still green,
"If"... Feel free to document that this is actually possible.
, then how can we rely on this?
Because the various dating methods give consistent results which have been repeatedly determined to be reliable and accurate.
Science goes out of its way to try to disprove the existance of God.
No, it really doesn't.
More so than to try and prove evolution.
You are extremely mistaken. There are multiple heavy monthly journals which consist of nothing but studies of evolution [one sample]. I can't think of a single article published in any peer-reviewed science journal which even attempted to "disprove the existence of God" (although you might find a few in the Philosophy department).
To deny a "supreme being" without proving "his" non-existance conclusively, is a fundemental error.
So... Since Shiva and Zeus and Odin haven't had their "non-existence conclusively proven", is it a "fundamental error" to deny them as well?
To prove "evolution" wihtout complete proof, such as all missing "links" is also an error.
You misunderstand how science works. Science does not deal in proofs.
However, by your own argument, if it's a "fundamental error" to "deny" something without "proving its non-existance conclusively", then aren't you making a "fundamental error" if you deny evolution without conclusively proving its falseness?
I think your thesis needs a bit more work. Meaning that the evidence, as more is gathered seems in direct contradiction to itself.
Feel free to present your alleged examples.
Leave anit-religious agendas out of science and deal with the facts found. But sadly it seems most science attacks religion
Speaking of agendas...
The Bible if nothing else, has proven things archeologically and including the existance of Peter. His house, and his name carved upon a stone. In the fishing village he was from. When you use the evidence found in the pages of the Bible and things start to add up, more truth upon more truth it is hard to deny the "whole" when the sum of its parts turn out to be real.
History and archeology teach us that the Civil War really happened, there really was a general named Sherman who burned Atlanta, there really were battles at certain places and times, etc., and that there was a woman named Margaret Mitchell. Does this make *all* of "Gone With The Wind" necessarily true?
Science has yet to find life on another planet. Yet they keep trying in this.
Because the only way to find out, *either way*, is to keep looking.
So they seem to have their own faith.
Yes, they have faith in the value of making efforts to keep learning more about the universe we live in.
But have yet to prove it to the world.
Nothing can be "proven" (there's that word again) without information. Science believes in gathering as much information as possible, so that when conclusions are made, they are based on real information, and not philosophical or religious dogmas or "sound-good-isms". And in gathering further information so that past conclusions can be further reality-checked. Science and the scienific method is, in a nutshell, all about doing frequent reality-checks of beliefs.
You misunderstand how science works. Science does not deal in proofs. Let's review what science actually is and what the scientific method actually does. (When one does, one realizes how far from science are conjectures of macroevolution, how far from scientifically validated are any descriptive hypotheses of how evolution is supposed to work, and how far from scientific theory is any patchwork model of the process of evolution.)
I'll present the information in roughly an order of very summarized to more detailed, so you can best decide where to stop.
1/4. from: http://www.soci.niu.edu/~phildept/Dye/method.html
Selected texts bolded in green, by unspun
Socratic Method and Scientific Method
| Socratic Method |
Scientific Method |
| 1. Wonder. Pose a question (of the "What is X ?" form). |
1. Wonder. Pose a question. |
| 2. Hypothesis. Suggest a plausible answer (a definition or definiens) from which some conceptually testable hypothetical propositions can be deduced. |
2. Hypothesis. Suggest a plausible answer (a theory) from which some empirically testable hypothetical propositions can be deduced. |
| 3. Elenchus ; "testing," "refutation," or "cross-examination." Perform a thought experiment by imagining a case which conforms to the definiens but clearly fails to exemplify the definiendum, or vice versa. Such cases, if successful, are called counterexamples. If a counterexample is generated, return to step 2, otherwise go to step 4. |
3. Testing. Construct and perform an experiment which makes it possible to observe whether the consequences specified in one or more of those hypothetical propositions actually follow when the conditions specified in the same proposition(s) pertain. If the experiment fails, return to step 2, otherwise go to step 4. |
| 4. Accept the hypothesis as provisionally true. Return to step 3 if you can conceive any other case which may show the answer to be defective. |
4. Accept the hypothesis as provisionally true.Return to step 3 if there other predictable consequences of the theory which have not been experimentally confirmed. |
| 5. Act accordingly. |
5. Act accordingly. |
Copyright © 1996, James Dye
Last Updated 8 January, 1996
2/4. from: http://www.ldolphin.org/SciMeth2.html

Steps in the Scientific Method
by Lambert Dolphin
Email: lambert@ldolphin.org
Web Pages: http://ldolphin.org/ May 1992.
3/4. from:
http://teacher.nsrl.rochester.edu/phy_labs/AppendixE/AppendixE.html
Selected texts bolded in green, by unspun
APPENDIX E: Introduction to the Scientific Method
The scientific method is the process by which scientists, collectively and over time, endeavor to construct an accurate (that is, reliable, consistent and non-arbitrary) representation of the world.
Recognizing that personal and cultural beliefs influence both our perceptions and our interpretations of natural phenomena, we aim through the use of standard procedures and criteria to minimize those influences when developing a theory. As a famous scientist once said, "Smart people (like smart lawyers) can come up with very good explanations for mistaken points of view." In summary, the scientific method attempts to minimize the influence of bias or prejudice in the experimenter when testing an hypothesis or a theory.
1. Observation and description of a phenomenon or group of phenomena.
2. Formulation of an hypothesis to explain the phenomena. In physics, the hypothesis often takes the form of a causal mechanism or a mathematical relation.
3. Use of the hypothesis to predict the existence of other phenomena, or to predict quantitatively the results of new observations.
4. Performance of experimental tests of the predictions by several independent experimenters and properly performed experiments.
If the experiments bear out the hypothesis it may come to be regarded as a theory or law of nature (more on the concepts of hypothesis, model, theory and law below). If the experiments do not bear out the hypothesis, it must be rejected or modified. What is key in the description of the scientific method just given is the predictive power (the ability to get more out of the theory than you put in; see Barrow, 1991) of the hypothesis or theory, as tested by experiment. It is often said in science that theories can never be proved, only disproved. There is always the possibility that a new observation or a new experiment will conflict with a long-standing theory.
As just stated, experimental tests may lead either to the confirmation of the hypothesis, or to the ruling out of the hypothesis. The scientific method requires that an hypothesis be ruled out or modified if its predictions are clearly and repeatedly incompatible with experimental tests. Further, no matter how elegant a theory is, its predictions must agree with experimental results if we are to believe that it is a valid description of nature. In physics, as in every experimental science, "experiment is supreme" and experimental verification of hypothetical predictions is absolutely necessary. Experiments may test the theory directly (for example, the observation of a new particle) or may test for consequences derived from the theory using mathematics and logic (the rate of a radioactive decay process requiring the existence of the new particle). Note that the necessity of experiment also implies that a theory must be testable. Theories which cannot be tested, because, for instance, they have no observable ramifications (such as, a particle whose characteristics make it unobservable), do not qualify as scientific theories.
If the predictions of a long-standing theory are found to be in disagreement with new experimental results, the theory may be discarded as a description of reality, but it may continue to be applicable within a limited range of measurable parameters. For example, the laws of classical mechanics (Newton's Laws) are valid only when the velocities of interest are much smaller than the speed of light (that is, in algebraic form, when v/c << 1). Since this is the domain of a large portion of human experience, the laws of classical mechanics are widely, usefully and correctly applied in a large range of technological and scientific problems. Yet in nature we observe a domain in which v/c is not small. The motions of objects in this domain, as well as motion in the "classical" domain, are accurately described through the equations of Einstein's theory of relativity. We believe, due to experimental tests, that relativistic theory provides a more general, and therefore more accurate, description of the principles governing our universe, than the earlier "classical" theory. Further, we find that the relativistic equations reduce to the classical equations in the limit v/c << 1. Similarly, classical physics is valid only at distances much larger than atomic scales (x >> 10-8 m). A description which is valid at all length scales is given by the equations of quantum mechanics.
We are all familiar with theories which had to be discarded in the face of experimental evidence. In the field of astronomy, the earth-centered description of the planetary orbits was overthrown by the Copernican system, in which the sun was placed at the center of a series of concentric, circular planetary orbits. Later, this theory was modified, as measurements of the planets motions were found to be compatible with elliptical, not circular, orbits, and still later planetary motion was found to be derivable from Newton's laws.
Error in experiments have several sources. First, there is error intrinsic to instruments of measurement. Because this type of error has equal probability of producing a measurement higher or lower numerically than the "true" value, it is called random error. Second, there is non-random or systematic error, due to factors which bias the result in one direction. No measurement, and therefore no experiment, can be perfectly precise. At the same time, in science we have standard ways of estimating and in some cases reducing errors. Thus it is important to determine the accuracy of a particular measurement and, when stating quantitative results, to quote the measurement error. A measurement without a quoted error is meaningless. The comparison between experiment and theory is made within the context of experimental errors. Scientists ask, how many standard deviations are the results from the theoretical prediction? Have all sources of systematic and random errors been properly estimated? This is discussed in more detail in the appendix on Error Analysis and in Statistics Lab 1.
As stated earlier, the scientific method attempts to minimize the influence of the scientist's bias on the outcome of an experiment. That is, when testing an hypothesis or a theory, the scientist may have a preference for one outcome or another, and it is important that this preference not bias the results or their interpretation. The most fundamental error is to mistake the hypothesis for an explanation of a phenomenon, without performing experimental tests. Sometimes "common sense" and "logic" tempt us into believing that no test is needed. There are numerous examples of this, dating from the Greek philosophers to the present day.
Another common mistake arises from the failure to estimate quantitatively systematic errors (and all errors). There are many examples of discoveries
Another common mistake is to ignore or rule out data which do not support the hypothesis. Ideally, the experimenter is open to the possibility that the hypothesis is correct or incorrect. Sometimes, however, a scientist may have a strong belief that the hypothesis is true (or false), or feels internal or external pressure to get a specific result. In that case, there may be a psychological tendency to find "something wrong", such as systematic effects, with data which do not support the scientist's expectations, while data which do agree with those expectations may not be checked as carefully. The lesson is that all data must be handled in the same way.
Another common mistake arises from the failure to estimate quantitatively systematic errors (and all errors). There are many examples of discoveries which were missed by experimenters whose data contained a new phenomenon, but who explained it away as a systematic background. Conversely, there are many examples of alleged "new discoveries" which later proved to be due to systematic errors not accounted for by the "discoverers."
In a field where there is active experimentation and open communication among members of the scientific community, the biases of individuals or groups may cancel out, because experimental tests are repeated by different scientists who may have different biases. In addition, different types of experimental setups have different sources of systematic errors. Over a period spanning a variety of experimental tests (usually at least several years), a consensus develops in the community as to which experimental results have stood the test of time.
In physics and other science disciplines, the words "hypothesis," "model," "theory" and "law" have different connotations in relation to the stage of acceptance or knowledge about a group of phenomena.
An hypothesis is a limited statement regarding cause and effect in specific situations; it also refers to our state of knowledge before experimental work has been performed and perhaps even before new phenomena have been predicted. To take an example from daily life, suppose you discover that your car will not start. You may say, "My car does not start because the battery is low." This is your first hypothesis. You may then check whether the lights were left on, or if the engine makes a particular sound when you turn the ignition key. You might actually check the voltage across the terminals of the battery. If you discover that the battery is not low, you might attempt another hypothesis ("The starter is broken"; "This is really not my car.")
The word model is reserved for situations when it is known that the hypothesis has at least limited validity. A often-cited example of this is the Bohr model of the atom, in which, in an analogy to the solar system, the electrons are described has moving in circular orbits around the nucleus. This is not an accurate depiction of what an atom "looks like," but the model succeeds in mathematically representing the energies (but not the correct angular momenta) of the quantum states of the electron in the simplest case, the hydrogen atom. Another example is Hook's Law (which should be called Hook's principle, or Hook's model), which states that the force exerted by a mass attached to a spring is proportional to the amount the spring is stretched. We know that this principle is only valid for small amounts of stretching. The "law" fails when the spring is stretched beyond its elastic limit (it can break). This principle, however, leads to the prediction of simple harmonic motion, and, as a model of the behavior of a spring, has been versatile in an extremely broad range of applications.
A scientific theory or law represents an hypothesis, or a group of related hypotheses, which has been confirmed through repeated experimental tests. Theories in physics are often formulated in terms of a few concepts and equations, which are identified with "laws of nature," suggesting their universal applicability. Accepted scientific theories and laws become part of our understanding of the universe and the basis for exploring less well-understood areas of knowledge. Theories are not easily discarded; new discoveries are first assumed to fit into the existing theoretical framework. It is only when, after repeated experimental tests, the new phenomenon cannot be accommodated that scientists seriously question the theory and attempt to modify it. The validity that we attach to scientific theories as representing realities of the physical world is to be contrasted with the facile invalidation implied by the expression, "It's only a theory." For example, it is unlikely that a person will step off a tall building on the assumption that they will not fall, because "Gravity is only a theory."
Changes in scientific thought and theories occur, of course, sometimes revolutionizing our view of the world (Kuhn, 1962). Again, the key force for change is the scientific method, and its emphasis on experiment.
While the scientific method is necessary in developing scientific knowledge, it is also useful in everyday problem-solving. What do you do when your telephone doesn't work? Is the problem in the hand set, the cabling inside your house, the hookup outside, or in the workings of the phone company? The process you might go through to solve this problem could involve scientific thinking, and the results might contradict your initial expectations.
Like any good scientist, you may question the range of situations (outside of science) in which the scientific method may be applied. From what has been stated above, we determine that the scientific method works best in situations where one can isolate the phenomenon of interest, by eliminating or accounting for extraneous factors, and where one can repeatedly test the system under study after making limited, controlled changes in it.
There are, of course, circumstances when one cannot isolate the phenomena or when one cannot repeat the measurement over and over again. In such cases the results may depend in part on the history of a situation. This often occurs in social interactions between people. For example, when a lawyer makes arguments in front of a jury in court, she or he cannot try other approaches by repeating the trial over and over again in front of the same jury. In a new trial, the jury composition will be different. Even the same jury hearing a new set of arguments cannot be expected to forget what they heard before.
The scientific method is intricately associated with science, the process of human inquiry that pervades the modern era on many levels. While the method appears simple and logical in description, there is perhaps no more complex question than that of knowing how we come to know things. In this introduction, we have emphasized that the scientific method distinguishes science from other forms of explanation because of its requirement of systematic experimentation. We have also tried to point out some of the criteria and practices developed by scientists to reduce the influence of individual or social bias on scientific findings. Further investigations of the scientific method and other aspects of scientific practice may be found in the references listed below.
1. Wilson, E. Bright. An Introduction to Scientific Research (McGraw-Hill, 1952).
2. Kuhn, Thomas. The Structure of Scientific Revolutions (Univ. of Chicago Press, 1962).
3. Barrow, John. Theories of Everything (Oxford Univ. Press, 1991).
Send comments, questions and/or suggestions via email to
wolfs@nsrl.rochester.edu.
4/4. from:
http://phyun5.ucr.edu/~wudka/Physics7/Notes_www/node5.html
Next: What is the ``scientific Up: Introduction Previous: Overview
Science is best defined as a careful, disciplined, logical search for knowledge about any and all aspects of the universe, obtained by examination of the best available evidence and always subject to correction and improvement upon discovery of better evidence. What's left is magic. And it doesn't work. -- James Randi
It took a long while to determine how is the world better investigated. One way is to just talk about it (for example Aristotle, the Greek philosopher, stated that males and females have different number of teeth, without bothering to check; he then provided long arguments as to why this is the way things ought to be). This method is unreliable: arguments cannot determine whether a statement is correct, this requires
proofs.
A better approach is to do experiments and perform careful observations. The results of this approach are universal in the sense that they can be reproduced by any skeptic. It is from these ideas that the scientific method was developed. Most of science is based on this procedure for studying Nature.
Jose Wudka
9/24/1998
I can show countless shows, books, lectures that try to disprove the existance of a creator. And one scientific documentary that tried to explain away the miracles in the Bible. It was pathetic. Such as Moses was able to part the Red Sea not from some miracle but because it was really shallow and filled with sandbars and at the moment it was parted there was a wind anomaly that created a downward sustained gale that hit the surface of the water and pushed it to each side. This show aired on NBC. Just one example.
Religion does not cause science to not exist. Facts are facts. But science discounts religion, and faith is claimed to blind those to science. Yet that science cannot agree whether we are now as evidence suggests, currently in an ice age. Which has happened every 11,000 years give or take a few. Over 200 feet of ice and snow covering planes from WWII that should have been much closer to the surface suprised those looking for them. And an weather shack erected in the mid 50's was also buried under 200 feet of ice and snow.
No hole in the ozone. Hole in the ozone. Global warming, global cooling. Second hand smoke bad, not so bad.
Science cannot agree even with the same evidence available to those doing the studies. It is or it is not. It cannot be both. We are warmer or cooler. And a recent study proves we were hotter 1400 years ago with wilder flucuations in temp than today. So once again proof is disproved. Until the next study that is.
Carbon dating has been done on a tree that was about 50 years dead, 1 test said it was 1300 yrs old another said it was 23,000 yrs old. Same specimen, different results.
Science cannot agree on some of the things mentioned above. They are in conflict with each other on whether or not T-Rex was a scanvenger or hunter. They cannot say what color a dino was, but they say from their appearant surroundings at the time this is what they might have been??? Science today lacks not because there is not evidence here or there on this or that, but too much science lately is infiltrated with PCism's. The latest textbooks say on the subject of Global warming that if the ice caps melt the earth would be covered by water 5 miles deep. It is wrong but PC to leave it that way. A scientist on a local radio show was asked this question and his response was that any real scientist knows this is wrong but when asked why it should be left that way he claimed that the fear that it can incite can bring awareness to the problem of global warming. Yet when the recent study about the Dark Ages was brought to his attention he dicounted it in it entirely.
With people like this listed as experts, PC is dictating results or so it would seem. Now we see a DISC channel show on a bunch of Dino hunters creating a machine to see just how a Sabertooth killed its prey. They attached a recreated tooth to a set of steel jaws welded to a backhoe. Then tested it on a cow carcass. A lot of effort to prove a theory yet the hydraulics can outdo an animals jaw strength.
There is also a show on what new species might look like based on theories of what the earth will change into. Such as a metallic spider that "farms" the "last" remaining mammal to provide it with food. ?????
Science has done great things but it seems too much effort is spent gathering donations and grants than doing actual science.


