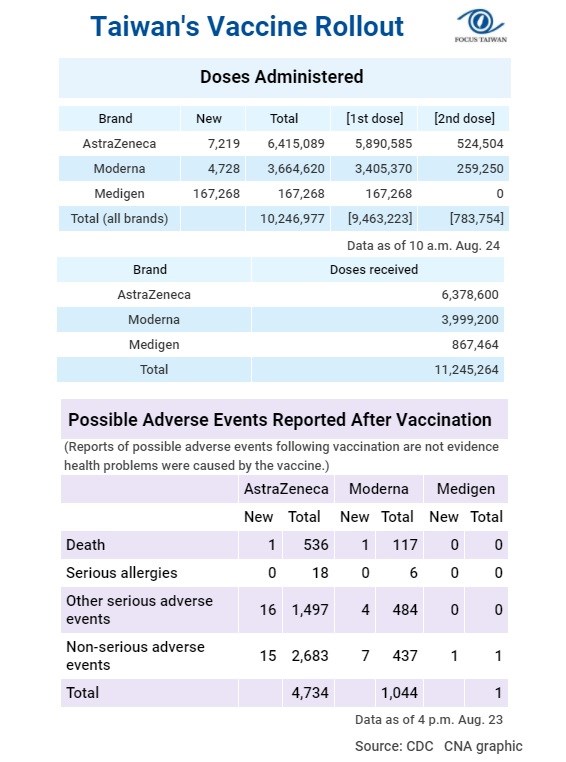
To: SeekAndFind
To: SeekAndFind
WHAT IS TAIWAN's MEDIGEN VACCINE?
The MVC COVID-19 vaccine, designated MVC-COV1901 and also known as the Medigen COVID-19 vaccine, is a protein subunit COVID-19 vaccine developed by Medigen Vaccine Biologics Corporation in Taiwan, American company Dynavax Technologies and the U.S. National Institute of Health.
This vaccine is made by the recombinant S-2P spike protein adjuvanted with CpG 1018 supplied by Dynavax. Preliminary results from Phase I trials on 77 participants were published in June 2021, indicating what the authors described as "robust" immune system response elicited by the vaccine.
On 13 October 2020, Medigen Vaccine Biologics received Taiwan's government subsidies for the initiation of Phase 1 Clinical Trial in Taiwan starting early October. The Phase 1 Clinical Trial was held at National Taiwan University Hospital with 45 participants ranging the age of 20–50.
On 25 January 2021, Medigen Vaccine Biologics initiated Phase 2 Clinical Trial for its COVID-19 vaccine candidate MVC-COV1901 with the first participant being dosed. The Phase 2 Clinical Trial for the MVC COVID-19 vaccine was a randomized, double-blinded, and multi-center clinical trial, planned to enroll 3,700 participants of any age 20 above.
On 10 June 2021, Medigen Vaccine Biologics released its COVID-19 vaccine Phase 2 interim analysis results, which demonstrates good safety profile in participants. The Phase 2 Clinical Trial in the end included 3,800 participants with all participants receiving second dose by 28 April 2021. Medigen Vaccine Biologics announced that it will request Emergency Use Authorization (EUA) with the concluding of the Phase 2 Clinical Trial.
On 20 July 2021, Medigen Vaccine Biologics filed a Phase 3 Clinical Trial IND application with Paraguay's regulatory authority, which was later approved. The Phase 3 Clinical Trial, however, was different from regular Phase 3 Clinical Trial, which uses immune-bridging trial to compare the performance of MVC COVID-19 vaccine with the Oxford-AstraZeneca COVID-19 vaccine.
On July 19, 2021, MVC COVID-19 vaccine obtained Emergency Use Authorization (EUA) approval from the Taiwanese government after fulfilling EUA requirements set by Taiwanese authority.
To: SeekAndFind
4 posted on
08/28/2021 7:12:42 PM PDT by
cmj328
(We live here.)
FreeRepublic.com is powered by software copyright 2000-2008 John Robinson