
Posted on 10/26/2020 6:48:07 AM PDT by SeekAndFind
There is renewed hope for elderly patients after tests showed a COVID-19 vaccine produced a similar immune response in both younger and older people.
The vaccine, being developed by the University of Oxford with drugs firm AstraZeneca, could be a crucial weapon in the battle to halt the spread of coronavirus.
Adverse responses to the vaccine were lower among the elderly, tests on the vaccine revealed.
Because the immune system weakens with age, it had been feared that the elderly might not respond as positively to the vaccine.
However, an AstraZeneca spokesman told the Reuters news agency: "It is encouraging to see immunogenicity responses were similar between older and younger adults and that reactogenicity was lower in older adults, where the COVID-19 disease severity is higher.”
Referring to the technical name of the vaccine, the spokesman added: “The results further build the body of evidence for the safety and immunogenicity of AZD1222.”
On Monday, the Financial Times reported that the vaccine triggers protective antibodies and T-cells in older age groups.
The vaccine is expected to be one of the first among the large pharmaceutical companies to gain regulatory approval, along with another being developed by Pfizer and BioNTech.
Immunogenicity blood tests carried out on a subset of older participants had similar results to data released in July that showed the vaccine generated "robust immune responses" in a group of healthy adults aged between 18 and 55, the Financial Times reported.
Health secretary Matt Hancock has said a vaccine will not be widely available until some time in the first half of next year, although he did not rule it out for NHS staff before Christmas.
(Excerpt) Read more at news.yahoo.com ...

As long as Biden doesn't win and claim credit for ending the virus, sheesh.
The discovery that the vaccine being developed by the University of Oxford, in collaboration with AstraZeneca, triggers protective antibodies and T-cells in older age groups has encouraged researchers as they seek evidence that it will spare those in later life from serious illness or death from the virus.
Age has emerged as the principal risk factor for a severe bout of Covid-19. However, the immune system weakens with age, raising concerns that the very group that most needs the protection of a vaccine may generate the least effective response to one.
People aware of the results from so-called immunogenicity blood tests carried out on a subset of older participants say the findings echo data released in July that showed the vaccine generated “robust immune responses” in a group of healthy adults aged between 18 and 55. The earlier findings showed that the vaccine induced two forms of human immune response — generating antibodies and T-cells — for at least 56 days, according to an analysis published in The Lancet.
In an infographic, the FT illustrates how the immune system responds to the virus.
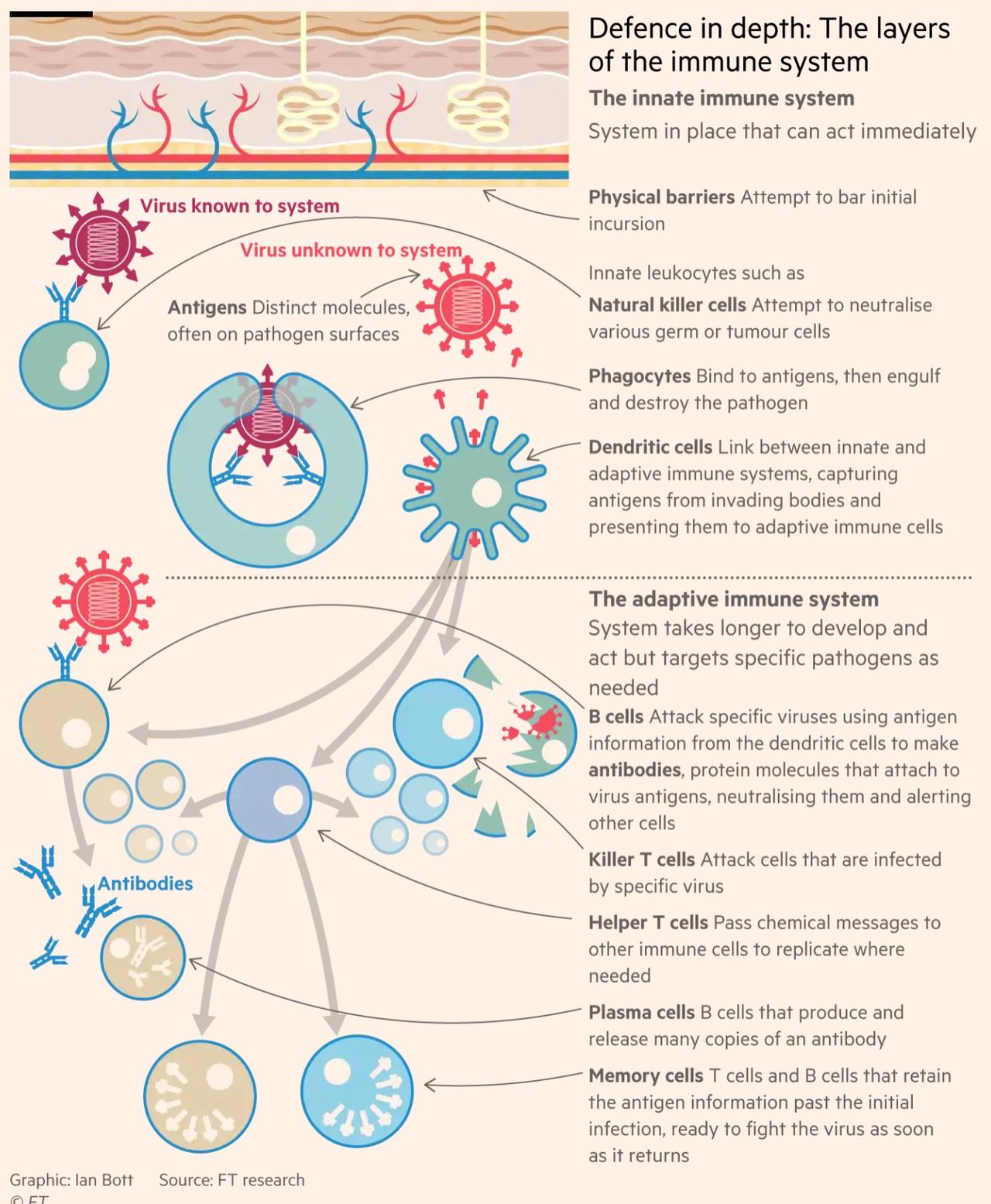
All very well to have a bureaucracy evaluating data on the safety and effectiveness of vaccines. In fact, IMHO Trump missed an opportunity when, after he asserted that vaccine will be available this year, the moderator asked, “Do you guarantee it?”Trump rightly admitted that he could not guarantee it. But he should, IMHO, have responded,
“What kind of question is that? I could only guarantee it if I were in charge of the bureaucracy which does those evaluations. I am not in charge of that - If I were, politically speaking, I could be distributing the vaccine right now because they are already in production in anticipation that at least one of them will be approved.The Democrats are irresponsiblly suggesting that if the bureaucracy does approve a vaccine that will be meaningless if it happens while I am president. I am proud of the fact that I have expedited everything else about the development of several different vaccines by different companies. But I have not, and do not want, any control over the evaluation of the safety and effectiveness of those developed vaccines. I say that a vaccine will be approved very soon, and I believe that. Because it’s not just one but several candidates being evaluated - and there is reason to suppose that all of those candidates will pan out. That’s not my call, it is the health care bureaucracy’s - but we have not put all our eggs in one basket and I think it’s 90% certain, at least, that vaccines will start being available this very year for those who deserve top priority in receiving them.”
In addition, I would remark that the question of safety and effectiveness is not absolute: theoretically it takes forever to absolutely prove safety or effectiveness. And the confidence level applied controls the speed at which approval can be given. And the more at-risk a population is, the less sense it makes to insist on a very high confidence level of safety of a vaccine which is clearly effective. If you really sharpened your pencil, you might conclude that the highest-risk population (apart from health care workers) is elderly/obese blacks - and that they should be offered the choice of taking the risk of the not-yet-fullly-proven vaccine instead of taking their chances without taking it.
Because as I said, all the vaccines are already in production for the simple reason that it is reasonable to believe that ultimately all of them will eventually prove out.
This is excellent information! Thank you for posting this. More people need to understand how all this works before they start forming (and expressing) opinions.
As usual, the devil is in the details.
Disclaimer: Opinions posted on Free Republic are those of the individual posters and do not necessarily represent the opinion of Free Republic or its management. All materials posted herein are protected by copyright law and the exemption for fair use of copyrighted works.