The "ULM neutron" in the diagram stands for "ultra low momentum neutron".

The "ULM neutron" in the diagram stands for "ultra low momentum neutron".
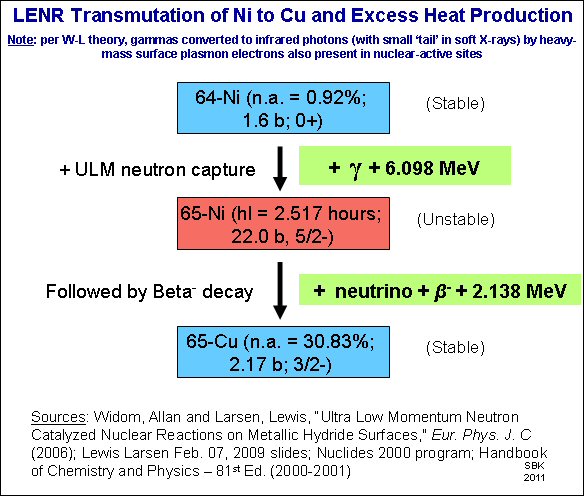
Explaining the diagram in my last post, 64-nickel (nickel with an atomic weight of 64, an isotope that occurs in 0.92% of natural nickel) captures an ultra-low-momentum neutron, and becomes 65-nickel (and emits energy). The 65-nickel has one of its neutrons turn into a proton (beta decay) emitting an electron (and more energy) and turning into 65-copper, a stable isotope.
Fascinating if it works. Conventional atomic physics focuses on forcing changes in an atom’s nucleus using direct means rather than manipulating the valence electrons.
This would be a rather civilized approach, coaxing transmutation via alteration of an atom’s electrons. But creating Cu from Ni should consume energy. The heat given off has to come from something decaying more than the energy required for the Cu creation.
Maybe this process was available all along but it does not provide what they were looking for, as in bomb before energy. If one is looking to create a bomb they are naturally going to be interested in the nucleus with a full association to its electron valence (maximum potential energy), i.e. cold isotopes.
This may explain why no one has pursued altering the valence electrons. I.e., why add a continual flow of energy to get a sustained reaction when you can add energy once to get a huge self-sustaining reaction? The nuclear energy field just followed-on using a less volatile fission decay process of the initial bombs.
I’ll just have to wait for CNN’s take on this..... < /s >