*************************************EXCERPT*************************************
The situation in the Gulf, in regard to the Deepwater Horizon fire and oil spill continues to evolve. Since my last post on this, the size of the operation has continued to grow, with some thought now being given to dredging barriers along the coastline. What I thought I cover a little today, among other things, is the problems that occur when oil leaves a hot reservoir and suddenly enters a pipe on the floor of a very cold sea, causing some of the constituent chemicals to crystallize out. To illustrate this, I took a picture of one of the pipes that used to sit in my office, since it helped visualize a problem we had been given, and is along the lines of that now facing BP.

You will note that the crystals have reduced the effective diameter of the pipe down to about 1.5 inches, cutting the flow the pipe can carry by 75% (roughly). These are but one example of a number of different chemicals that can precipitate out in the pipes that carry oil from the reservoir, where it is hot, up through colder zones as it rises through the different pipes on its way to land. There are two different aspects to the problem that I want to cover since it relates to the formation of methane hydrates in the capture box that BP had fielded at the end of last week.
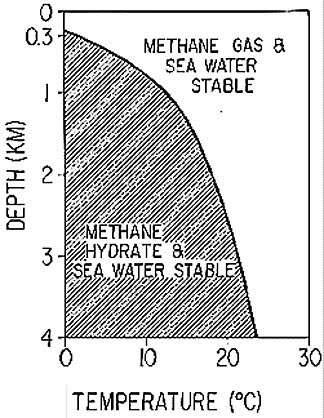
The first of these is on the growth of the crystal hydrates within the confinement box that BP had lowered over the middle leak in the riser. However, rather than dig back into my notes, I am going to suggest that those who want to understand the chemistry/physics go to The Obligate Scientist. He uses a plot from the USGS to illustrate the conditions that transition methane gas to methane hydrate as a function of temperature and pressure (depth in the ocean).