Skip to comments.
AIDS Infection Rate in U.S. Higher Than Previously Estimated
Washington Post ^
| August 2, 2008
| David Brown
Posted on 08/02/2008 12:22:48 PM PDT by neverdem
click here to read article
Navigation: use the links below to view more comments.
first previous 1-20 ... 61-80, 81-100, 101-120, 121-133 next last
To: GodGunsGuts
Duesberg, Raznick, Ellison and several others have offered to do just that. All they require is that the NIH/CDC properly supervise the experiment, confirm that they have been injected with HIV and test positive for the same, and regularly report on their progress thereafter. In one other instance, Raznick offered to have himself injected without the above caveat, if the scientist he was debating at the time would agree to go on AIDS chemotherapy. At first, the scientist accepted the challenge, but later backed out.Do you have something to confirm this assertion?
81
posted on
08/02/2008 10:28:54 PM PDT
by
neverdem
(I'm praying for a Divine Intervention.)
To: neverdem
==It depends on the country, drug availability and choice of patient given their situation. I’ve read a few studies results where the drug or drugs were given for a minimum of a day or two antenatally and intrapartum. It works.
You missed the whole point of my reply. Bottom line, AZT is extremely toxic and harms both mother and baby. The side effects are known, and they aren’t pretty. Even if you assume that HIV is the cause of AIDS, it only infects 1 in 500 T-cells. That means they have to kill 500 healthy cells just to get one infected cell. Is it any wonder there are so many nasty side effects? And if HIV is not the cause of AIDS, these treatments are downright criminal!
To: GodGunsGuts
83
posted on
08/02/2008 10:35:17 PM PDT
by
count-your-change
(you don't have to be brilliant, not being stupid is enough.)
To: neverdem
==Do you have something to confirm this assertion?
Duesberg et al have issued many such offers, and I have no inclination to track them all down. The following will have to suffice. If you want to track the rest of them down, be my guest.
The following is from a rebuttal written by Duesberg and Ellison and published in the (conservative) Hoover Institution’s “Policy Review”:
But both of us would be quite willing to carry out the Fumento test: if he will arrange for sufficient national publicity, if he would be convinced by our action, and if he will thereafter help us bring exposure to our viewpoint, we will indeed be quite happy to have ourselves publicly injected with HIV. Perhaps Fumento will also be willing to check on our health status in the year 2000, or after whatever additional time is eventually added to the virus’s latent period.*
http://www.duesberg.com/about/pdpolicyrep.html
You can read the original article that prompted the rebuttals here:
http://www.duesberg.com/about/pdpolicy.html
To: count-your-change
To: GodGunsGuts
To: Alamo-Girl
Have you been following this at all?
To: GodGunsGuts
That's a really bogus offer. All he has to do is inject himself and become HIV positive and let the infection run it's course. or non-course.
But if you want the last word you're welcome to it.
88
posted on
08/02/2008 11:45:46 PM PDT
by
count-your-change
(you don't have to be brilliant, not being stupid is enough.)
To: count-your-change
No, I think I will let you have the LAST word, C-Y-C. If you ever actually sit down, roll up your sleeves, and make a sincere attempt to understand BOTH sides, be sure and give me a ping. I say both sides, because it’s clear you don’t even understand your side of the debate, let alone my side. Cheers—GGG
To: GodGunsGuts; All
90
posted on
08/03/2008 12:38:28 AM PDT
by
neverdem
(I'm praying for a Divine Intervention.)
To: hinckley buzzard
You're right, of course.
I would have better made my point with a question mark, instead of a period, or used a /sarcasm tag.
91
posted on
08/03/2008 4:07:03 AM PDT
by
G.Mason
(Duty, Honor, Country)
To: GodGunsGuts
“But as Duesberg notes, while AZT lowers transmission of HIV from mother to child by 17%, it comes at the cost of putting 100% of those same mothers on AZT CHEMOTHERAPY.”
But what explanation does Duesberg have for AZT lowering transmission of HIV from mother to child? What mechanism does he propose that avoids having AZT acting as an anti-retroviral? (inasmuch as Douchberg insists it is Chemotherapy).
92
posted on
08/03/2008 7:41:34 AM PDT
by
allmendream
(If "the New Yorker" makes a joke, and liberals don't get it, is it still funny?)
To: xDGx
“If HIV isn’t the cause of AIDS, then what is?”
Advanced liberalism.
93
posted on
08/03/2008 7:50:08 AM PDT
by
wgflyer
(Liberalism is to society what HIV is to the immune system.)
To: neverdem
Those charts actually make your point, after screening began AIDS cases went up and then began to drop, as would be expected with a disease that showed symptoms some time after first infection. Or would we infer from that drug use went down due to screening? I think the former.
That so-called long after was only a few years which is exactly what we should see when the causative agent is in the blood. Hepatitis C, also passed in blood transfusions, also might infect person with no symptoms for months or even years later. Hence after tests were developed in the early 90’s a “look program was initiated. So those charts actually support the point that HIV causes AIDS.
94
posted on
08/03/2008 8:22:52 AM PDT
by
count-your-change
(you don't have to be brilliant, not being stupid is enough.)
To: neverdem
An article pertinent to your post appeared in the Aug.3 Las Vegas Sun. Might be worth a read if you’ve time.
95
posted on
08/03/2008 8:47:02 AM PDT
by
count-your-change
(you don't have to be brilliant, not being stupid is enough.)
To: neverdem; count-your-change
==Those charts actually make your point, after screening began AIDS cases went up and then began to drop, as would be expected with a disease that showed symptoms some time after first infection.
Wrong again. They began screening ALL blood products for HIV in 1985. According to the CDC, "Nearly all people infected with HIV through blood transfusions received those transfusions before 1985, the year HIV testing began for all donated blood."
In addition, how do you explain the following? Pay attention now...
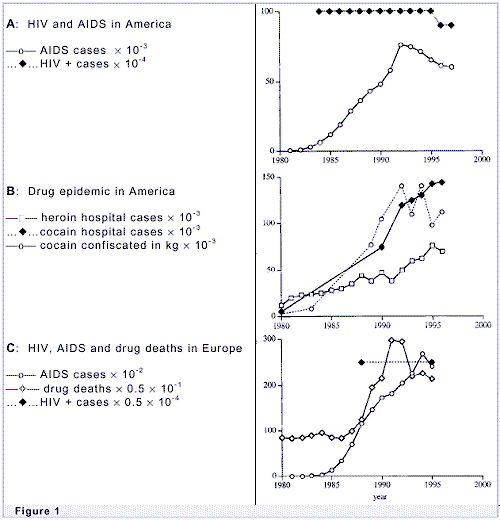
To: allmendream
==But what explanation does Duesberg have for AZT lowering transmission of HIV from mother to child? What mechanism does he propose that avoids having AZT acting as an anti-retroviral? (inasmuch as Duesberg insists it is Chemotherapy).
Let me begin with the your last comment first. If AZT is not a chemotherapy drug, what is it?
As for your questions, I’ve never heard Duesberg’s explanation as to the mechanism. I can only assume he thinks it’s because AZT is a chain-terminator of DNA that prevents all cells from dividing. If you prevent all cells from dividing, then you reduce the number of HIV infected T-cells from dividing, thus lowering the transmission of HIV from mother to child.
To: xDGx
I first took samples of blood that, at 1:400 dilution, tested negative for antibodies to HIV. I then ran the exact same serum samples through the test again, but this time without diluting them. Tested straight, they all came positive. And that's supposed to be an argument against the accuracy of the tests?
If one wanted to be confident in the accuracy of the tests, a good protocol would probably be to run each test on samples diluted 100x, 200x, 400x, 800x, and 1600x, and see which samples test positive. If people who seem to have HIV were to consistently test positive on the 800x (or even 1600x) samples while those without HIV were to consistently test negative on the 200x (or even 100x) samples, that would be a pretty strong indication that the phenomenon being measured was significant enough that a single sample at 400x would be sufficient to detect it reliably.
If the test requires dilution 400x, that would suggest that the inherent goal in the test is to measure, quantitatively what degree of dilution is required for a negative result. If 400x is sufficient, the patient is HIV negative; if not, the patient is HIV positive.
98
posted on
08/03/2008 10:49:08 AM PDT
by
supercat
To: GodGunsGuts
You missed the whole point of my reply. Bottom line, AZT is extremely toxic and harms both mother and baby. The side effects are known, and they aren’t pretty. Even if you assume that HIV is the cause of AIDS, it only infects 1 in 500 T-cells. That means they have to kill 500 healthy cells just to get one infected cell. Is it any wonder there are so many nasty side effects? And if HIV is not the cause of AIDS, these treatments are downright criminal!Au contraire! I'm a family practice doc with a special interest in adverse drug reactions because of my personal experience, except it wasn't with AZT, a phony nucleic acid intended to interfere with the normal functions of the RNA of the virus and what should be the expected blowback on host DNA and RNA.
AZT isn't the only drug against HIV/AIDS. There are at least 20 of them having 6 different ways of thwarting the virus. For me, the proof is in the pudding, i.e. increased longevity and decreased mortality of those infected with HIV which causes AIDS. Please read the links in comment# 90.
99
posted on
08/03/2008 12:15:34 PM PDT
by
neverdem
(I'm praying for a Divine Intervention.)
To: GodGunsGuts
AZT’s mechanism in HIV and AIDS suppression is chain termination, but what about drugs that specifically target reverse transcriptase? Moreover as you can see in this study, a specific mutation in reverse transcriptase makes HIV resistant to AZT as it will no longer accept is as an analog to Adenosine.
http://jvi.asm.org/cgi/content/abstract/82/7/3261
We identified clinical isolates with phenotypic resistance to nevirapine (NVP) in the absence of known nonnucleoside reverse transcriptase inhibitor (NNRTI) mutations. This resistance is caused by N348I, a mutation at the connection subdomain of human immunodeficiency virus type 1 (HIV-1) reverse transcriptase (RT). Virologic analysis showed that N348I conferred multiclass resistance to NNRTIs (NVP and delavirdine) and to nucleoside reverse transcriptase inhibitors (zidovudine [AZT] and didanosine [ddI]). N348I impaired HIV-1 replication in a cell-type-dependent manner. Acquisition of N348I was frequently observed in AZT- and/or ddI-containing therapy (12.5%; n = 48; P < 0.0001) and was accompanied with thymidine analogue-associated mutations, e.g., T215Y (n = 5/6) and the lamivudine resistance mutation M184V (n = 1/6) in a Japanese cohort. Molecular modeling analysis shows that residue 348 is proximal to the NNRTI-binding pocket and to a flexible hinge region at the base of the p66 thumb that may be affected by the N348I mutation. Our results further highlight the role of connection subdomain residues in drug resistance.
100
posted on
08/03/2008 12:22:41 PM PDT
by
allmendream
(If "the New Yorker" makes a joke, and liberals don't get it, is it still funny?)
Navigation: use the links below to view more comments.
first previous 1-20 ... 61-80, 81-100, 101-120, 121-133 next last
Disclaimer:
Opinions posted on Free Republic are those of the individual
posters and do not necessarily represent the opinion of Free Republic or its
management. All materials posted herein are protected by copyright law and the
exemption for fair use of copyrighted works.
FreeRepublic.com is powered by software copyright 2000-2008 John Robinson
