Skip to comments.
Anthrax Vaccine Safe, Effective, Health Chief Says
DefenseLink ^
| DefenseLink
Posted on 12/26/2003 10:16:44 PM PST by xzins
Anthrax Vaccine Safe, Effective, Health Chief Says
By Jim Garamone American Forces Press Service
WASHINGTON -- With almost a million shots given, the anthrax immunization is proving to be one of the safest vaccination program on record, said Dr. Sue Bailey, assistant secretary of defense for health affairs.
“The vaccine that we are administering to our troops for protection against anthrax is effective and entirely safe,” Bailey said during a Pentagon interview.
She said service members are experiencing few serious adverse reactions from the shots. The most recent reports show only 14 reactions were serious enough that the service members had to be off work for 24 hours or more or hospitalized, she said. “Those who had those kinds of reactions have fully recovered,” she added.
As of June 16, 935,632 separate shots had been administered. With 102 total reactions reported, including the 14 serious reactions, this means only .01 percent of the shots caused an adverse reaction. “This is a lower rate of reaction than one gets with a [diptheria, pertussis, tetanus] shot administered to children,” Bailey said.
Bailey countered reports that the vaccine was somehow tainted with a substance called squalene. Squalene is a substance that appears naturally in everyone’s body, she explained. “You also find it in a lot of beauty products and in some health food products,” she said. “But, squalene has never been used in the anthrax immunization vaccine production, and it is not now present.”
Following the reports, DoD contracted with a civilian laboratory that tested the vaccine for squalene and “found there is no squalene in the anthrax vaccine we are using,” she said.
Bailey said the vaccine DoD uses is effective. “[Anthrax is] so deadly, we don’t test humans,” she said. “We rely upon non-human primate testing to give us the information about the efficacy of the vaccine. And that shows it to be very effective in protecting against anthrax.”
The anthrax vaccine the department uses is licensed by the Food and Drug Administration and has been since 1970, Bailey said. The vaccine stocks have undergone DoD-mandated supplemental testing performed by the manufacturer and overseen by a private, independent firm.
Since 1970, there have been no reports of long-term adverse health effects from the anthrax vaccine. Howerver, DoD continues to study the vaccine. “We have a study underway at the U.S. Army Medical Insitute of Infectious Diseases to determine whether individuals who received multiple vaccines, including anthrax, demonstrate any adverse health effects over the long term,” Bailey said. DoD has another study underway at Tripler Army Medical Center in Hawaii. A total of 570 medical workers who have received the series are being studied so DoD can get “on-going information that we can project into the future about effects of the anthrax vaccine.”
The anthrax vaccination program is a series of six shots stretching over 18 months. DoD started immunizing service members most in danger from anthrax -- those in or going to Southwest Asia. In May 1998, Defense Secretary William S. Cohen approved a plan to inoculate all service members against the disease.
“Anthrax is a deadly bacteria,” Bailey said. “If you were exposed to weaponized anthrax spores and were not immunized, you would develop symptoms and die. Antibiotics alone cannot save you once you display the symptoms. We feel it is our responsibility to provide for the best protection … and we do so through the immunization program.”

Surveillance for Adverse Events Associated with Anthrax Vaccination --- U.S. Department of Defense, 1998--2000
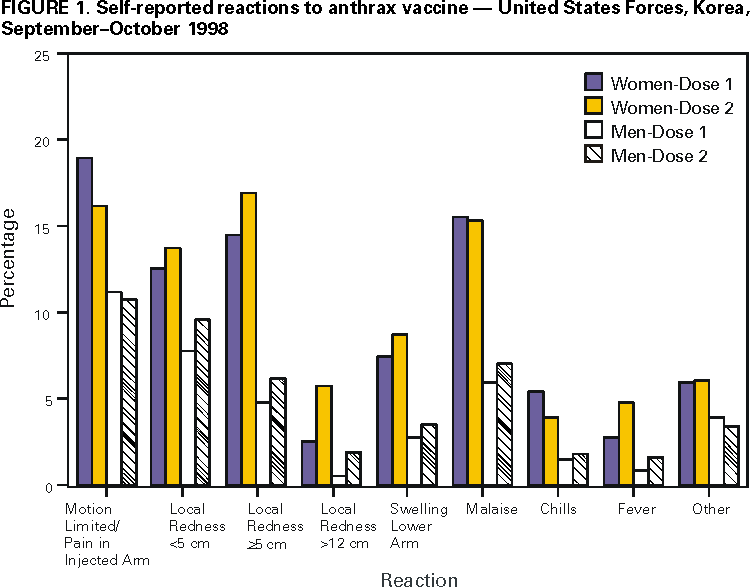
Concerns about the potential use of anthrax as a biologic weapon prompted the U.S. Department of Defense (DoD) to announce on December 15, 1997, anthrax vaccination of all U.S. military personnel. This effort is coordinated by the Anthrax Vaccine Immunization Program (AVIP). AVIP plans a phased vaccination process to achieve total force protection against anthrax by 2004. The current phase of implementation includes vaccination of all service members and mission-essential DoD civilian employees assigned or deployed to high-threat areas. On the basis of program monitoring, as of April 12, 2000, 425,976 service members had received 1,620,793 doses of anthrax vaccine adsorbed (AVA) (Bioport, Inc.,* Lansing, Michigan). Some service members have cited concerns about vaccine safety and efficacy in their decision to refuse vaccination, despite the possibility of administrative or disciplinary actions. To assess anthrax vaccination safety, DoD has conducted surveys of vaccinated personnel. This report describes three completed or ongoing surveys (1). The findings indicate that rates of local reactions were higher in women than men and that no patterns of unexpected local or systemic adverse events have been identified. Survey of Self-Reported Reactions to AVA, U.S. Forces, Korea
At one of the largest vaccination sites for United States Forces, Korea, a mandatory, self-administered prevaccination questionnaire was used to obtain data on health status (including pregnancy, if applicable), medication use, and reactions to the previous dose of AVA. The questionnaire was designed to record service members' concerns about AVA and their reports of adverse events (i.e., a medical condition following vaccination that could be related to the vaccine) to promote risk communication between health-care providers and service members. Data from 6879 questionnaires completed during September--October 1998 were reviewed. Approximately 37% (2531 of 6879) of respondents were service members receiving their first dose; records were analyzed for 4348 (63%) service members who already had received and could comment on their first (2427) or second (1921) vaccine doses. Female service members reported higher rates of reactions to the previous dose of vaccine, regardless of the time period after vaccination (Figure 1). For both men and women, most reported that events were localized, minor, self-limited, and did not lead to impaired work performance, lost work time beyond that required to seek care, and/or a clinic visit or hospitalization. After the first or second dose, 82 (1.9%) of 4348 reported that their work performance had been limited to some extent or that they were placed on limited duty, 13 (0.3%) reported <1 day lost from work, 21 (0.5%) consulted a clinic for evaluation, and one (0.02%) required hospitalization for an injection site reaction. Tripler Army Medical Center Survey of AVA Safety
Tripler Army Medical Center, Honolulu, Hawaii, assessed the frequency and nature of AVA adverse events in a cohort of 603 U.S. military health-care workers in the Korea Medical Augmentee Program. These personnel began receiving anthrax vaccination during September 1998. A self-administered questionnaire was used to collect data prospectively for systemic reactions. Data on local reactions were collected retrospectively for the first three doses and prospectively for the remaining doses. Persons responded to questions on symptoms, signs, time taken off from duty, hospitalizations, and medical visits. Medical records were reviewed and information was obtained from health-care providers about participants who sought medical care, missed one or more work shifts, or had any reaction that might exempt them from further vaccination. Data collection up to the fourth AVA dose of the six-dose initial series was complete for 479 (79.4%) of 603 persons. Of the remaining 124 (20.6%), 11 were not vaccinated because of pregnancy, four were exempted from the survey for medical reasons, and the rest were lost to follow-up primarily because of reassignment. After the first anthrax dose, 47 (7.9%) of 595 reported seeking medical advice and/or taking time off work for a complaint (e.g., muscle or joint aches, headache, or fatigue); after the second dose, 30 (5.1%) of 585; after the third dose, 16 (3.0%) of 536; and after the fourth dose, 17 (3.1%) of 536. Vaccine Adverse Events Reporting System (VAERS)
DoD uses the CDC and Food and Drug Administration (FDA) Form VAERS-1 to report events potentially related to any vaccination to VAERS and to each military service's disease reporting system. VAERS reports related to anthrax vaccinations are consolidated for AVIP by the Defense Medical Surveillance System. As of April 7, 2000, 428 VAERS-1 reports had been received through DoD. Of these, 311 (72.7%) concerned systemic reactions, 78 (18.2%) were reports on mild or moderate local reactions, and 39 (9.1%) were for large or complicated local reactions. Thirty-six (8.4%) reactions met the DoD mandatory reporting criteria (i.e., hospitalization and/or time off duty >24 hours). None were related to suspected vaccine lot contamination. A panel of civilian scientific and medical experts established by the U.S. Department of Health and Human Services at DoD's request reviewed all VAERS-1 reports, including those reported directly to FDA or CDC. As of March 21, 2000, the panel has not identified any unexpected patterns of adverse events among 674 reports reviewed. Reported by: PA Sato, MD, M Ryan, MD, GC Gray, MD, Naval Health Research Center, San Diego, California. KJ Hoffman, MD, CN Costello, MD, United States Forces, Korea. GM Wassermann, MD, Tripler Army Medical Center, Honolulu, Hawaii. MV Rubertone, MD, SA Stanek, DO, US Army Center for Health Promotion and Preventive Medicine, Aberdeen Proving Grounds, Maryland. JD Grabenstein, PhD, Anthrax Vaccine Immunization Program Agency, Office of the Army Surgeon General, Falls Church, Virginia. JR Riddle, DVM, D Trump, MD, Office of the Assistant Secretary of Defense, US Dept of Defense, Washington, DC. Editorial Note:
Anthrax is considered a biologic weapons threat because of its stability in spore form, its ease of culture, the absence of natural immunity in industrialized nations, and severity of infection in its gastrointestinal and inhalational forms. If untreated, the case-fatality rate of inhaled anthrax exceeds 80% (2,3). At least seven nations are suspected to have actively pursued biologic weapons programs (3,4). Anthrax also has been used at least once by terrorist groups (3,4). U.S. service members deployed to future military confrontations may be at risk for attack by biologic warfare agents. The DoD, through the AVIP, seeks to reduce these threats. Human anthrax vaccine was licensed by FDA in 1970 as a six-dose primary series with annual boosters. It is an aluminum hydroxide-adsorbed, cell-free, noninfectious vaccine prepared from a noncapsulating, nonproteolytic anthrax strain. Licensing was based on safety data, the results of a controlled efficacy trial, and observational data documenting substantial protection against anthrax (5,6). Studies in nonhuman primates also have documented protection (7). The safety and efficacy of this vaccine was affirmed by an independent advisory panel in 1985 (5). This mandatory vaccination program has posed substantial challenges to DoD. Some service members are reluctant to be vaccinated because of concern about adverse events. These concerns may be heightened by the number of doses required and by allegations linking vaccination with illnesses in Gulf War veterans. Conversely, some service members may not report adverse events after vaccination because of concerns that they will not be able to complete the vaccination series, thereby limiting career advancement options. The findings in this report provide information on rates of local and systemic adverse events occurring after anthrax vaccination was delivered as part of a routine program rather than in clinical trials. The findings suggest that rates of local reactions were higher in women than men and that no patterns of unexpected local or systemic adverse events have been identified. Reasons for the higher rates in women are unknown. The studies reported here are subject to several methodologic limitations, including sample size, the power to detect rare adverse events, loss to follow-up, and exemption of vaccine recipients with previous adverse events. For example, in the U.S. Forces, Korea, study, any service members medically deferred after a previous AVA dose would have been missed by the survey; therefore, adverse events may have been underreported. In the Tripler survey, data were collected retrospectively for the first three doses and then prospectively, potentially resulting in recall or observational bias. In addition, in the Tripler survey, the absence of an unvaccinated control group limited the ability to assess an association of adverse events with anthrax vaccination. The studies were not designed to detect or quantify chronic or long-term adverse events. Ongoing activities at DoD, CDC, and FDA are targeted toward improving methods to communicate the benefits and risks for vaccination, enhancing surveillance for vaccine adverse events, and continuing to monitor the safety of the program. These interventions may be useful to enhance AVIP. Risk-communication programs, such as the one in U.S. Forces, Korea, encourage a positive and supportive patient-provider relationship. Surveillance through the VAERS system to detect signals of potential adverse events followed by epidemiologic investigations to evaluate these signals remains important. Potential methodologies for monitoring safety include comparing vaccinated and unvaccinated groups or comparing groups shortly after vaccination with groups whose vaccinations were more distal. Pilot studies have evaluated intramuscular vaccine administration to reduce rates of local adverse events. Additional studies are planned to expand these data and to determine whether the number of doses required in the primary vaccination series can be reduced while maintaining immunogenicity and protection. AVIP maintains a World-Wide Web site (http://www.anthrax.osd.mil)† with information on the program and electronic mail access to AVIP staff. A toll-free information line for inquiries from health-care providers, service members, and the public also is available (telephone [877] 438-8222). References
- Friedlander AM, Pittman PR, Parker GW. Anthrax vaccine: evidence for safety and efficacy against inhalational anthrax. JAMA 1999;282:2104--6.
- Brachman PS, Friedlander AM. Anthrax. In: Plotkin SA, Orenstein WA, eds. Vaccines. 3rd ed. Philadelphia, Pennsylvania: WB Saunders, 1999.
- Inglesby T, Henderson D, Bartlett J, et al. Anthrax as a biological weapon: medical and public health management. JAMA 1999;281:1735--45.
- Mazzuchi JF, Claypool RG, Hyams KC, et al. Protecting the health of U.S. military forces: a national obligation. Aviation, Space, and Environmental Med 2000;71:260--5.
- Food and Drug Administration. Biological products; bacterial vaccines and toxoids: implementation of efficacy review. Federal Register 1985;50:51002--117.
- Brachman PS, Gold H, Plotkina SA, Fekety FR, Werrin M, Ingraham NR. Field evaluation of a human anthrax vaccine. Amer J Pub Hlth 1962;52:432--45.
- Dixon TC, Meselson M, Guillemin J, Hanna PC. Medical progress: anthrax. N Engl J Med 1999;341:815--26.
* Use of trade names and commercial sources is for identification only and does not imply endorsement by CDC or the U.S. Department of Health and Human Services. † References to sites of non-CDC organizations on the Internet are provided as a service to MMWR readers and do not constitute or imply endorsement of these organizations or their programs by CDC or the U.S. Department of Health and Human Services. CDC is not responsible for the content of pages found at these sites.
Figure 1
Return to top. |
TOPICS: Anthrax Scare; Extended News; Foreign Affairs; Miscellaneous; News/Current Events; War on Terror
KEYWORDS: anthrax; anthraxvaccine; effectiveness; immunization; spore; vaccine
1
posted on
12/26/2003 10:16:44 PM PST
by
xzins
To: xzins
I don't necessarily see a big problem with the vaccine....but the method of production might fail every test. Consider this scenario...you want a vaccine in a hurry which normally is produced at very low levels (even the vet's out in west Texas admitted that they didn't know anyonewho took the stuff). So you the Pentagon put this out for a bid...and only Bioport bids (out 200 potential companies)...no one else even submitted a bid. They turn around within weeks and start mass producing the stuff...and problems start to arise with the people receiving the vaccine. Five years later...yes, five....then FDA comes around to Bioport and does a inspection of the facility. It fails. If this had been Bayer...it would be a huge episode, with people being fired. Bioport promptly announces that they don't have enough money to fix the facility and that DOD must pay for this. DOD hands over millions because this is the only company willing to produce the stuff (again, the only company). Not only do they fix their production center....but heck, they even buy brand new furniture for the facility, and even pave the parking lot....all with your Government funding.
No one in congress wants to investigate this matter...and the amusing thing is the court system appears to be the only arm of the government willing to take on this whole matter. This would make a great Groucho Marx movie....except alot of sick GIs are at the heart of the matter. It would seem like that the best target in America for Ossama to hit...would be Bioport...and no one else would stand up to produce this garbage.
To: pepsionice
Kudos to #1. I was hoping I was not the only following the Bioport government handout. Remember, a retired general was hired by Bioport prior to the vaccine being manufactured, supposedly to help run Bioport. However, we all know how the retired brass going into businesses that deal with the government reek of favortism. This smells of something rotten all the way from the top down in the Clinton administration.Somebody saw a golden goose and the military personnel have been the golden egg to Bioport.
To: xzins
This is the Pentagon propaganda put out in 1999 before they got their pee-pees handed to them by GAO, APHA, and Congress. (
http://www.ngwrc.org/anthrax/timeline.html) A brief background on the military's Anthrax Vaccine Immunization Program
by Kathryn D. Hubbell
1. The current anthrax vaccine was licensed base on data from a different vaccine.
See anthrax vaccine chronology on House Government Reform web site at
http://www.house.gov/reform 2. There are no studies of human safety and efficacy regarding this vaccine. The only study which can be quoted, known as the Brachmann Study, was actually done on the original vaccine, not the re-configured vaccine which has been used by the military on its troops. Other studies quoted by BioPort, the vaccine's manufacturer, have not been peer-reviewed or published.
See anthrax vaccine chronology on House Government Reform web site at
http://www.house.gov/reform 3. BioPort, the current manufacturer of the vaccine, has a long and troubled history with the manufacture of the product, including:
· Producing contaminated batches - and using them
· Producing non-sterile product - and using it
· Switching labels on batches of the vaccine to reveal false expiration dates
See anthrax vaccine chronology on House Government Reform web site at
http://www.house.gov/reform for these three bullets
· · Using equipment for other vaccines besides the anthrax vaccine, in direct violation of FDA rules
· Failing to have a written protocol to maintain consistency between batches and lots
See the FDA Citizen Petition at
http://www.ngwrc.org/anthrax for these three bullets
2. There was never any rationale for the six-shot protocol used with the vaccine. The original protocol was for three shots, and the change was made without study or scientific backing.
See anthrax vaccine chronology on House Government Reform web site at
http://www.house.gov/reform 3. The vaccine has always been known to be highly reactogenic, and the new label on the vaccine admits as much.
Where the old label claimed an adverse reaction rate of just 0.2%, the new label says adverse reactions may be as high as 35%.
Where the old label admitted to no fatalities, the new label describes the six deaths related to the vaccine.
The new label also details a far stronger list of adverse reactions than did the old label.
See
http://www.fda.gov/cber/label/biopava0131022LB.pdf 6. After last fall's anthrax attacks via the U.S. Mail, civilians were offered the anthrax vaccine in combination with antibiotic drugs known to be effective against anthrax, such as Cipro and Doxycycline. They were offered informed consent documents, as required by law, since the anthrax vaccine has for years been on file with the FDA as an Investigational New Drug. However, no member of the U.S. military has ever been offered informed consent documents. The Centers for Disease Control (CDC) claims that civilians were offered informed consent documents because the vaccine was given post-exposure, rather than pre-exposure. However, this explanation circumvents the fact that the vaccine is still on IND status with the FDA, and the FDA has never issued a final ruling on that status. See
http://www.ngwrc.org/anthrax/CDCdocs.shtml 7. BioPort was suddenly cleared by the FDA to start manufacturing the vaccine again after last fall's anthrax attacks, after many years of failing FDA inspections. If they are manufacturing according to the old formula, then we can still expect a high adverse reaction rate with more permanent illnesses and more fatalities. If they have once again re-configured the vaccine without notifying the FDA or other authorities, then the vaccine is once again - still - an Investigational New Drug, and constitutes medical experimentation on the troops. In the brief eight or nine months since BioPort's clearance to resume the manufacturing process, there certainly has not been enough time to develop and test a new drug. In fact, recent news of a second generation anthrax vaccine clearly states that it may take until 2009 for such a drug to be developed, tested and licensed, unless it is offered earlier as an Investigational New Drug. See
http://ebird.dtic.mil/Aug2002/s20020808dod.htm 8. The Department of Defense announced in early July, 2002, that it would resume the Anthrax Vaccine Immunization Program within the military, but only for those in undefined "high risk areas," who were deployed there more than 15 days, and only in complete secrecy. The original announcement included the fact that no troop who received the vaccine would be allowed to talk about it, leading one to wonder what those troops are expected to do when they become ill from the vaccine, as some of them certainly will. Prohibiting any talk of the vaccine also leads the Department of Defense to be able to deny a connection between the vaccine and a subsequent illness, or cluster of illnesses - as has already happened with Gulf War veterans, whose shots were never entered into their medical records.
In the same announcement, the Department of Defense announced with it was withholding half of its stockpiled vaccine - and by "stockpiled," one has to ask whether this is the vaccine that was quarantined at BioPort as being unfit for use - for civilian use. They want to share this vaccine with the civilian population. See
http://story.news.yahoo.com/news?tmpl=story&u=/ap/20020517/ap_wo_en_ge/us_anthrax_vaccine_3 The headline on the above article reads, "Military wants to limit troops' anthrax vaccinations; question of civilian needs unanswered," which leads us to the next point:
9. Wisconsin is among those states which have now passed a law declaring that in time of national emergency, taking vaccines such as the anthrax vaccine will be mandatory. Under the Homeland Security Act, various states are joining Wisconsin in considering mandatory bioterrorism vaccines for the general public. These include (but are not limited to): Maine ("Two homeland security bills sent to governor -House clarifies bioterror attack powers," By PATRICK JACKSON - Dover Bureau reporter, The News Journal, 06/27/2002), Georgia (Story last updated at 2:28 p.m. on Sunday, January 20, 2002, "Online Athens," "Terror attacks spur new security laws" By Brian Basinger, Morris News Service), and Oklahoma ("Public health, eavesdropping top lists as states craft anti-terror policies" By the Associated Press, Aug. 13, 2002, in "Shawnee Online.")
Meanwhile, we have veterans in their 20's walking with canes, crippled with arthritic conditions since taking the anthrax vaccine; we had tough former Marines and Army Special Forces who have grand mal seizures, blackouts, and severe memory loss since taking the vaccine; women who are unable to conceive, and hemorrhage constantly; men whose bodies will no longer produce testosterone; troops who have lost vision in one eye, or peripheral vision; who have blood clots, brain tumors, cysts on internal organs, severe skin rashes and infections, short-term memory loss, depression and dementia, and diagnosed autoimmune illnesses - all since taking the vaccine. Nearly every person who has reported becoming ill since the vaccine expresses complete mystification and dismay that they have changed so completely from being physically fit, athletic, and active, to a person whose disabilities prevent any kind of a normal life.
The Veterans Administration, in July of 2002, issued a precedent-setting ruling recognizing a disability specifically related to the anthrax vaccine, the same week the Department of Defense announced a resumption of the shots. See
http://www.va.gov/ogc/docs/PREC_4-2002.doc This is a major step forward, because since the shots were first given (as far back as the 1970's), troops have been repeatedly told there is no connection to their illnesses and the anthrax vaccine; and if they can prove there is at all, they most often fight one-to-two years to obtain even partial VA benefits. One result is that they often go bankrupt in the process as they face the dual stress of mounting medical bills while they are no longer able to earn a living. Many of our veterans - our nation's finest, in their healthier days - have become homeless, and have had to move back in with family members or friends.
The Department of Defense has long stated that some 16 bioterrorism vaccines are in the pipeline, and has many times expressed concern that an AIDS vaccine be developed. An AIDS vaccine is now being tested in Thailand, for that matter. It is of some note that the adjuvant squalene, used in several experimental vaccines and found by the FDA to be present in various lots of the anthrax vaccine, is also an adjuvant considered for the AIDS vaccine. Recent research out of Tulane University points to squalene antibodies present in those who have received the anthrax vaccine, and points to a definitive link between the anthrax vaccine and the cluster of conditions and illnesses known as Gulf War Syndrome. See
http://www.autoimmune.com/NewsRel15July02.html, and
http://www.autoimmune.com/GWSGen.html In addition, a recent study by Kansas State University found, among other things, that "Declines in long-term subjective health were associated with receipt of anthrax vaccine by Gulf War veterans but not for those who did not deploy to the Gulf, although few of the latter received anthrax vaccine." (cover page), and that "While the U.S. military maintains that 'There have been no long-term side effects from the vaccine,' (footnote, page 8) our research and Unwin, et al's (footnote, page 36) suggest to the contrary that anthrax vaccination might have direct and indirect long-term side effects on health, most notably for those individuals who have the most severe reactions to the vaccinations initially." (page 651)
Contact head researcher: Dr. W. R. Schummm, School of Family Studies and Human Services, Justin Hall, Kansas State University, 1700 Anderson Ave.,Manhattan, KS 66506-1403, or e-mail to:
Schumm@humec.ksu.edu
4
posted on
12/26/2003 11:03:07 PM PST
by
optimistically_conservative
(Nothing is as expensive as a free government service or subsidized benefit.)
To: optimistically_conservative
I know Dr Schumm....I got my master's degree out of his department at KSU. (He's a good, very Christian man.)
In any case, here's the link for an article from the Journal of the American Medial Assoc. It's in some kind of locked pdf so I can only give the link.
Jama and Inhalational Anthrax, click
If someone knows how to post the article itself, I'd appreciate it.
5
posted on
12/27/2003 7:23:56 AM PST
by
xzins
(Retired Army and Proud of It!)
To: optimistically_conservative; xzins
Haven't I read that there is no evidence that this vaccine has any efficacy at all against inhalation anthrax? If that's true, then I don't see the point of subjecting our soldiers to any risk at all through the vaccine, since inhalation anthrax is presumably the form of anthrax that is the threat to them.
To: aristeides
Go to the link at #5. The interesting thing about that review is that it's in the Journal of the American Medical Association....prestigious.
There are animal tests that say the vaccine is not effective against guinea pigs. However, when they went up to primates, particularly the rhesus monkey, they had very good success against inhalation anthrax.
There was one study on cutaneous anthrax with some mill workers that showed good results in protection. Interestingly, some of those same mill workers (both placebo and immunized) sadly were exposed to inhalation anthrax. Those who had received the immunization fared better.
Read the Jama study. Since DoD is standing by their research, this is not a simple "bad military, bad DoD, bad president, all trying to kill our soldiers" issue. DoD and the president have a lot to lose if they force this on troops and KNOW they don't have any leg to stand on.
7
posted on
12/27/2003 2:24:20 PM PST
by
xzins
(Retired Army and Proud of It!)
To: xzins; aristeides; optimistically_conservative; taxesareforever; pepsionice
Another roadkill on the road to force protection:
http://www.kpvi.com/index.cfm?page=nbcheadlines.cfm&ID=16047 LOCAL SOLDIER GIVEN ANTHRAX VACCINE
12/26/2003
Wednesday, a U.S. district judge ordered the military to stop adminstering the anthrax vaccine to service members without their consent until a trial can be held on the matter. Plaintiffs in the case argue that the anthrax vaccine is being used as an experimental drug.
But for one local Air Force member, the ruling may have come too late. Bryan Latham explains.
Vance Wasden, USAF-honorably discharged:
"Last Christmas Eve, they had to have an ambulance come and get me because my wife thought that I was dying because I stopped breathing and my eyes rolled in the back of my head."
It is just one of the many traumas Vance Wasden lives through. But it wasn't always like this - Wasden was once a very healthy 24-year-old serving in the United States Air Force. However, his life changed back in 1998 when he had an adverse reaction to the anthrax vaccine.
"The joint pain, the muscular pains, the memory loss, concentration problems..."
Plus chronic fatigue and fibromyalgia - all which he battles daily. In total, Wasden lives with over a dozen different conditions - all due, he says, to a bad vaccine.
"My vaccination was five years old and they changed the date on my vaccine just so they could give it to me and the FDA inspections and Congressional oversight also proved sequeline was in my vaccine illegally."
Suprisingly, Wasden says he whishes he could still serve, loves the military, and has nothing but admiration for those still
serving.
"I am not doing this out of hostility towards the military. I am just hoping that what I say and what other people say will protect our troops."
He has a document outlining other members of his unit who have become sick too.
"I hope that they will take the time to write their congressmen, write their senator, and write to Washington, and ask that the people who were jailed - they'll be given an honorable discharge, and all those who are sick be given a proper military retirement and for the families of those people who are dead, will get retribution."
What he wants is every soldier to be properly honored.
More than 800,000 troops have received the anthrax vaccine since 1998.
8
posted on
12/30/2003 12:21:33 AM PST
by
zipper
(Retired Air Force Officer, still fighting for soldiers' rights)
Disclaimer:
Opinions posted on Free Republic are those of the individual
posters and do not necessarily represent the opinion of Free Republic or its
management. All materials posted herein are protected by copyright law and the
exemption for fair use of copyrighted works.
FreeRepublic.com is powered by software copyright 2000-2008 John Robinson
