
Posted on 02/17/2023 7:41:40 PM PST by DoodleBob
...Combining results from a community-wide serological survey of 5,310 blood donors and volunteers with SARS-CoV-2 viral load data from city-wide wastewater surveillance, the researchers estimated, (i) vaccine effectiveness against all Omicron BA.2 infections conferred by two, three and four homologous doses of the Comirnaty or CoronaVac vaccines for 100 days after each dose and (ii) COVID-19 infection attack rate in Hong Kong from 1 January to 31 July 2022. The researchers developed two in-house ELISA assays detecting IgG antibodies to the nucleocapsid (N) or Open Reading Frame 8 (ORF8) protein of SARS-CoV-2, with the latter assay developed specifically to detect past infection in CoronaVac vaccines.
The researchers estimated three and four doses of Comirnaty were 48% and 69% effective in preventing Omicron infection, respectively, 7 days after vaccination, waning to 26% and 35% by 100 days after vaccination. Three and four doses of CoronaVac were 30% and 56% effective after 7 days respectively, declining to 6% and 11% by 100 days.
(Excerpt) Read more at alphagalileo.org ...
Per the FDA in June 2020, the FDA would expect that a COVID-19 vaccine would prevent disease or decrease its severity in at least 50% of people who are vaccinated.
This is where words become REALLY important. In the US, the basis of approval EUA and the only "approved" Vaccine, was for PREVENTION...NOT for decreased severity. Indeed, look at the actual FDA and Pfizer-BioNTech info.
On August 23, 2021, the FDA approved the first COVID-19 vaccine. The vaccine has been known as the Pfizer-BioNTech COVID-19 Vaccine, and will now be marketed as Comirnaty, for the prevention of COVID-19 disease in individuals 16 years of age and older.(emphasis added)
Per the Comernity Insert,
--------------------------- INDICATIONS AND USAGE----------------------------
COMIRNATY is a vaccine indicated for active immunization to prevent coronavirus disease 2019 (COVID-19) caused by severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) in individuals 16 years of age and older. (1)(emphasis added)
Per the Summary Basis for Regulatory Action, under "Indication"
Active immunization to prevent coronavirus disease 2019 (COVID-19) caused by severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) in individuals 16 years of age and older. (Emphasis added)
Clearly, the vaccine is for PREVENTION. Nothing else.
And four doses of Comirnaty can barely do that after three months. In fact, the VE is below the FDA's 50% threshold.
Approval should be revoked.

One of the myriad reasons I left that shithole. HK just marketed the Biontech as Comirnity, which has never been mass produced.
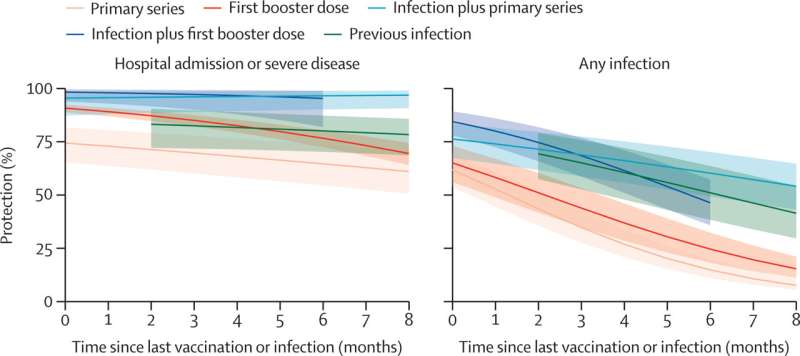
What is interesting in the above graphs is that natural immunity gives much better protection than the vax. Only the primary series of shots allegedly improves it (hybrid immunity). The booster shots actually reduce your protection if you already have natural immunity. For some reason the article fails to point this out.
Do not take the booster if you already have natural immunity. It will actually weaken your immune system if you do.
Four shots and 3 months later you’re practically back to square one? For a version of the virus which is really not threatening to most people. The versions that are floating around now are not worth sticking yourself with any vaccine. Even a real vaccine.
I’m 71 my wife is 69 and our doctor told us that we are at very little risk because we are in good health. Don’t worry about, it just go about your life. We both got some version of it last February. We survived, it wasn’t terrible. There will be no vaccines of any kind in our house ever at this point in our lives.
Gotta hand it to Pfizer. AFAIK, they have never claimed the China bioweapon is long lived. Neither are the vaccinated.
What the fork?
Getting no shot works even better
https://www.alphagalileo.org/en-gb/AlphaGalileo/About-us
An "Economy: subscription is a mere $1,320.00 for "academics" and a bargain "Standard" $2,650.00 subscription for business.
Thus, reading a "for free" article is a matter of their intent. But here's the thing. No survey is written as this: "...the researchers estimated...." Used more than once, and then my favorite, "The researchers expected..."
Thinking plainly, "vaccine effectiveness waned rapidly thereafter over 100 days" means three jabs a year for "effectiveness."
Against a variant which is already eight months ago, with new variants being touted by the jab industry.
Oddly, the article did not speak to deaths per se with data and numbers, merely with assertions. This is marketing. Three jabs a year marketing. But it's dressed up prettily with the HK crest and all.
The FDA approval in August 2021 was for licensing of the vaccine as Comirnaty. The FDA will not give final approval until all of the trials have been completed. There are 11 trials, the last of which is scheduled to be concluded in 2025. You’ll know when/if the vaccine receives final approval when it is available America. At that point, Big Pharma will be liable for any vaccine injuries and the EUA will be canceled.
To all the Big Pharma advocates here on FR. Time for another fix.
Disclaimer: Opinions posted on Free Republic are those of the individual posters and do not necessarily represent the opinion of Free Republic or its management. All materials posted herein are protected by copyright law and the exemption for fair use of copyrighted works.