|
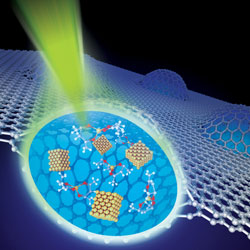
The graphene cover slip allows researchers to watch liquid chemistry taking place in much greater detail
© Image courtesy of Alivisatos, Lee and Zettl research groups, Lawrence Berkeley National Laboratory and KAIST
|
Posted on 04/08/2012 12:54:35 PM PDT by neverdem
The list of remarkable applications for graphene grows ever longer. This time, scientists in the US and Korea have shown that the single-atom thick carbon membrane can be used as a cover slip for an electron microscope to allow atomic-resolution observations of wet chemistry - something that is notoriously tricky to achieve.

|
The graphene cover slip allows researchers to watch liquid chemistry taking place in much greater detail
© Image courtesy of Alivisatos, Lee and Zettl research groups, Lawrence Berkeley National Laboratory and KAIST
|
Zettl and his colleagues figured that graphene might make for a good liquid cell. 'Graphene is continuous without holes, it is very flexible and very strong, and best of all it is only one atom thick, and it is very transparent to the electron beam,' Zettl says.
The team fabricated two layers of graphene and trapped a solution of platinum ions between them, creating a blister of graphene enclosing the solution. In the microscope, the incident electrons reduce the platinum ions to the metal, to form nanocrystals. The graphene liquid cell allowed the researchers to observe crystal growth in unprecedented detail. 'The formation shows more active faces attracting new atoms, and the merging of small crystallites into larger ones,' says Zettl. 'Different models have been proposed for the growth, but now we know which ones are correct by direct observation. It is a bit like seeing a finished ancient Egyptian temple and then proposing models of which blocks were put down in which order; but now we can watch the temple being built block by block. It gives us tremendous new insight.' The technique should be applicable to a wide range of liquid-phase systems, Zettl says: 'Basically, any wet chemistry.'
Valeria Nicolosi, who uses high spatial resolution electron microscopy to study two-dimensional nanomaterials, including graphene, at the University of Oxford, UK, is impressed by the work. 'I find this approach of using graphene as a vehicle for in situ liquid cell imaging revolutionary and innovative to say the least,' she says. 'They not only created an environment which realistically reproduces non-perturbative conditions for the liquid specimen, allowing in situ live investigation, but allowed atomic-resolution imaging and minimised heating effects under the electron beam.'
J M Yuk et al, Science, 2012, 336, 61 (DOI: 10.1126/science.1217654)
BTW, wet chemistry just means a chemical reaction in a liquid solution. There's a short video at the Chemistry World source. I can't capture the embeded code for Youtube.
It would be nice if this technology were applied to cancer research. We may at long last find a cure for it.
I worked with a pharmaceutical crystallization that was a 36 hour batch process and the crystal product formed had two stereo isomers, a right hand and left hand but identical in all physical/chemical respects otherwise. However, only the right hand was active as a therapeutic with the left hand being inert. A better understanding of the crystallization chemistry could yield a greater purity of the active right hand isomer and thus reduce production cost followed by reduced retail cost to the patient. By the way, production cost of this pharmaceutical chemical was several thousand dollars per pound.
Let's pick an ammonia fertilizer as an industrial crystallization with production costs of I don't know, say a buck pound. Purity is not near as strong a driver because the economic value of 95% versus 99%+ does not have as much value to the customer, the farmer in the field. It does though have a significant impact to the manufacturer as one aspect of the reduced purity are inclusions of the saturated reaction fluid trapped within pockets within the crystal lattice (imperfections). Lost reaction fluid means greater raw material costs to mix up the reaction fluid. Finally the point! LOL! If this basic research can shed light on what process variables favor or disfavor inclusion of reaction fluid in the crystal lattice then process factors can be adjusted minimize loss of reaction fluid and thus reduce production cost. Ultimately, this would mean more dollars to shareholders and lower cost to the farmer.
Graphene seems to be so useful in things that are uneconomical or impossible now that in the future we will talk about it as new era, before graphene/after graphene.
Thanks for the post. It’s an exciting time to be alive.
I was thinking the same thing. The stuff is like the discovery of plastic.
I can already envision some great in vacuo experiments -- such as observing micro-corrosion in real time with a SEM. Almost makes me want to go back to work...
Almost... '-}
Disclaimer: Opinions posted on Free Republic are those of the individual posters and do not necessarily represent the opinion of Free Republic or its management. All materials posted herein are protected by copyright law and the exemption for fair use of copyrighted works.