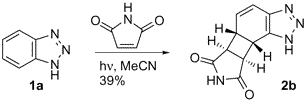
Light is used by chemists to catalyze a wide variety of reactions that are thermally forbidden, and yes, most of these are irreversible. Previously these reactions used to be done just by setting the reaction flask in the windowsill or on the benchtop. Now we have apparatuses that produce the optimal frequences, but the principle is the same. Here is an example:
This is a nice example because it is an intermolecular reaction resulting in a product that is very highly functionalized.
"Photochemistry of Benzotriazole: An Unprecedented Tautomer-Selective Intermolecular [2+2] Photocycloaddition." Booker-Milburn, K.; Wood, P.; Dainty, R.; Urquhart, M.; White, A.; Lyon, H.; Charmant, J. Organic Letters, 2002, 4, 1487-1489.
Obviously then light can initiate such reactions and objections on Second Law grounds are not in keeping with reality.